- EN - English
- CN - 中文
A Rapid and Visual Soybean Hairy Root Transformation Protocol Using the RUBY Reporter
发布: 2026年03月20日第16卷第6期 DOI: 10.21769/BioProtoc.5635 浏览次数: 10
评审: Ying LiAnonymous reviewer(s)
Abstract
Agrobacterium rhizogenes–mediated hairy root transformation provides a rapid platform for gene function analysis prior to stable whole-plant transformation. However, most existing hairy root transformation methods rely on tissue culture and require chemical or fluorescence-based selection, which increases experimental complexity. Here, we describe a tissue culture–free soybean hairy root transformation protocol incorporating the RUBY visual reporter system. While this work does not introduce a new transformation concept, it presents a streamlined implementation of established soybean hairy root methodologies that emphasizes procedural simplicity, reduced handling, and faster access to functional root material. Transgenic roots expressing RUBY can be directly identified by red pigmentation with the naked eye. In RUBY-positive roots, candidate genes driven by the CaMV 35S promoter showed higher expression levels than those in empty-vector controls, indicating that the system supports effective gene expression. Using this procedure, clearly identifiable transgenic hairy roots can be obtained within 20 days. Overall, this protocol simplifies induction and screening while reducing operational complexity and equipment requirements.
Key features
• Induces soybean hairy roots without tissue culture, simplifying the transformation workflow.
• Incorporates RUBY reporter for direct naked-eye identification of transgenic roots.
• Generates identifiable transgenic hairy roots within 20 days, enabling rapid functional analysis.
• Applicable to gene and protein function studies, metabolite profiling, and root–microbe interaction assays.
Keywords: Agrobacterium rhizogenesGraphical overview
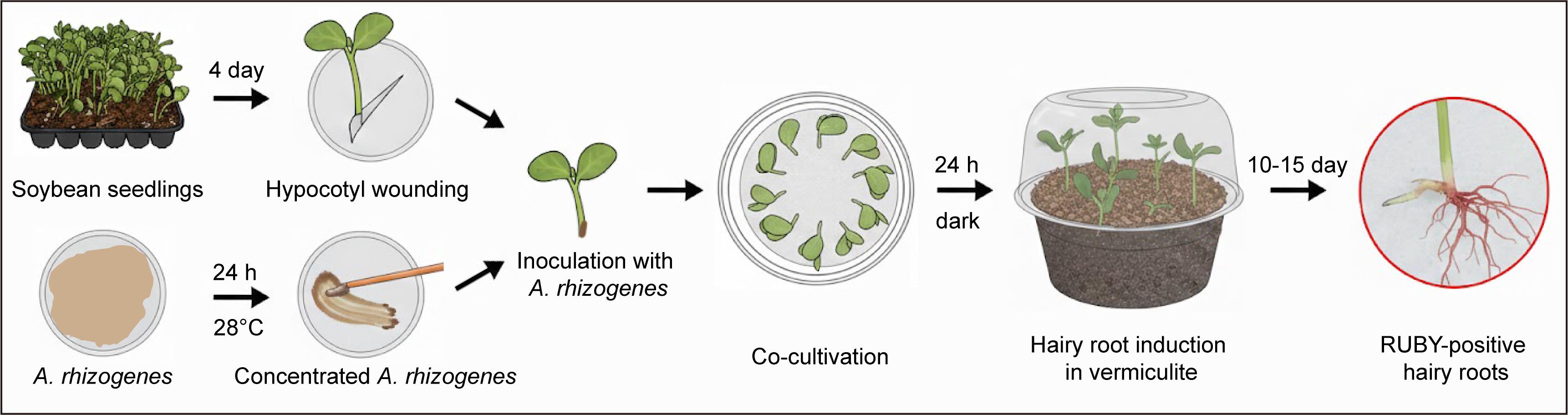
Schematic diagram illustrating the workflow of soybean hairy root transformation using the RUBY reporter
Background
Agrobacterium rhizogenes is a gram-negative soil bacterium belonging to the family Rhizobiaceae that infects plants through wound sites. During infection, transfer DNA (T-DNA) carried on the Ri plasmid is delivered into plant cells and randomly integrated into the host genome, where the introduced genes are expressed and induce the formation of roots with sustained growth capacity [1,2]. Because this process bypasses the requirement for whole-plant regeneration, A. rhizogenes–mediated hairy root transformation enables rapid generation of root tissues with stable transgene expression.
Hairy root transformation has therefore become a widely used experimental system in plant functional genomics. The approach is applicable to diverse molecular and physiological studies, including protein expression, subcellular localization, and protein–protein interaction analyses [3,4], secondary metabolism pathway investigation [5,6], and root–microbe interaction studies [7,8]. In addition, hairy root systems have been increasingly applied in genome editing research as a convenient platform for evaluating CRISPR/Cas9 single-guide RNA (sgRNA) efficiency, with editing outcomes that show good consistency with stable transformation results [8,9].
In soybean [Glycine max (L.) Merr.], conventional whole-plant stable transformation is time-consuming, labor-intensive, genotype-dependent, and often exhibits low efficiency, which limits its application in rapid gene function analysis and high-throughput studies [10,11]. A. rhizogenes–mediated hairy root transformation overcomes many of these limitations, providing an efficient alternative for root-focused studies, including analyses of metabolism, abiotic stress responses, and plant–microbe interactions [12–16]. Nevertheless, many existing soybean hairy root protocols still involve tissue culture steps and require antibiotic selection or fluorescence microscopy to identify positive transformants, with overall experimental durations typically ranging from 30 to 45 days [17,18].
To address these limitations, we built upon a tissue culture–free soybean hairy root induction system previously reported by Chen et al. [19] and incorporated the RUBY visual reporter gene for direct identification of transgenic roots. RUBY is a synthetic reporter comprising three betalain biosynthetic enzymes, CYP76AD1, DODA, and glucosyltransferase, which together drive the accumulation of a vivid red pigment in transformed cells [20,21]. Because RUBY expression can be readily detected by the naked eye, it provides an effective indicator for transformation events and transgene expression levels. Incorporation of the RUBY reporter eliminates the need for antibiotic or fluorescence-based selection, simplifies experimental procedures, and reduces equipment requirements, thereby offering a streamlined and reproducible method for generating transgenic soybean hairy roots suitable for downstream functional analyses.
Materials and reagents
Biological materials
1. Soybean seeds (Williams 82)
2. Agrobacterium rhizogenes K599 (Coolaber Science & Technology, catalog number: CC410)
3. E. coli DH5α competent cell (Sangon Biotech, catalog number: B528413)
4. pCAMBIA1300 plasmid (CAMBIA, stored in our laboratory)
Reagents
1. Xho I restriction enzymes (New England Biolabs, catalog numbers: R0146); store at -20 °C
2. Kanamycin sulfate (Sangon Biotech, catalog number: A600286); store at 4 °C
3. Streptomycin sulfate (Sangon Biotech, catalog number: A610494); store at 4 °C
4. Tryptone (OXOID, catalog number: LP0042B); store at room temperature (RT)
5. Yeast extract (OXOID, catalog number: LP0021T); store at RT
6. Sodium chloride (NaCl) (Sangon Biotech, catalog number: A501218); store at RT
7. Calcium chloride dihydrate (CaCl2·2H2O) (Sangon Biotech, catalog number: A501330); store at RT
8. Glycerol (Sangon Biotech, catalog number: A600232); store at RT
9. Sodium hypochlorite (NaClO) (Sangon Biotech, catalog number: A501944); store at 4 °C
Caution: NaClO is a strong oxidizing agent.
10. Hydrochloric acid (HCl), 37% (Sigma-Aldrich, catalog number: 320331); store at RT
Caution: HCl is highly corrosive.
11. Acetosyringone (AS) (Beijing KEHBIO Technology, catalog number: K91068); store at 4 °C
12. Agar powder (Beijing Solarbio Science & Technology, catalog number: A8190); store at RT
13. KOD-Plus DNA polymerase (TOYOBO, catalog number: KOD-201); store at -20 °C
14. EZ-10 Spin Column DNA Gel Extraction kit (Sangon Biotech, catalog number: B610353); store at RT
15. Agarose (Sangon Biotech, catalog number: A620014); store at RT
16. ClonExpress MultiS One Step Cloning Kit (Vazyme Biotech, catalog number: C113); store at -20 °C
17. Dimethyl sulfoxide (DMSO) (Diamond, catalog number: A100231); store at RT
Caution: DMSO can facilitate dermal absorption of chemicals and may cause skin irritation.
18. Tris base (Sangon Biotech, catalog number: A501492); store at RT
19. Glacial acetic acid (Sinopharm Chemical Reagent, catalog number: 10000208); store at RT
20. EDTA disodium salt dihydrate (Sangon Biotech, catalog number: A610185); store at RT
Solutions
1. Liquid and solid lysogeny broth (LB) medium (see Recipes)
2. 0.1 M NaCl solution (see Recipes)
3. 20 mM CaCl2 solution (see Recipes)
4. AS stock solution (see Recipes)
5. Kanamycin stock solution (see Recipes)
6. Streptomycin stock solution (see Recipes)
7. 50× TAE buffer (see Recipes)
Recipes
1. Liquid and solid LB medium
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Tryptone | 10 g/L | 10 g |
| NaCl | 10 g/L | 10 g |
| Yeast extract | 5 g/L | 5 g |
| Agar powder (for solid LB only) | 15 g/L | 15 g |
| Deionized water | n/a | Up to 1L |
Note: Autoclave the medium at 121 °C for 15 min and store at RT for up to one month.
2. 0.1 M NaCl solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| NaCl | 0.1 M | 5.84 g |
| Deionized water | n/a | Up to 1 L |
Note: Filter-sterilize the solution using a 0.22 μm syringe filter and store at 4 °C for six months.
3. 20 mM CaCl2 solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| CaCl2·2H2O | 20 mM | 0.29 g |
| Deionized water | n/a | Up to 100 mL |
Note: Filter-sterilize the solution using a 0.22 μm syringe filter and store at 4 °C for six months.
4. AS stock solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| AS | 100 mg/mL | 1 g |
| DMSO | n/a | Up to 10 mL |
Note: Filter-sterilize the solution using a 0.22 μm syringe filter, aliquot into 500 μL portions, and store at -20 °C and protected from light for up to six months.
5. Kanamycin stock solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Kanamycin sulfate | 50 mg/mL | 2.5 g |
| Deionized water | n/a | Up to 50 mL |
Note: Filter-sterilize the solution using a 0.22 μm syringe filter, aliquot into 500 μL portions, and store at -20 °C for six months.
6. Streptomycin stock solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Streptomycin sulfate | 50 mg/mL | 2.5 g |
| Deionized water | n/a | Up to 50 mL |
Note: Filter-sterilize the solution using a 0.22 μm syringe filter, aliquot into 500 μL portions, and store at -20 °C for six months.
7. 50× TAE buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Tris base | 2.0 M | 242 g |
| Glacial acetic acid | 1.0 M | 57.1 mL |
| EDTA (disodium salt, pH 8.0) | 50 mM | 100 mL of 0.5 M EDTA |
| Deionized water | n/a | Up to 1 L |
Note: Dilute 50× TAE buffer to 1× with deionized water before use. Store 50× stock at RT for six months.
Laboratory supplies
1. Glass dishes (12 cm dish) (WuYi)
2. Filter paper, 11 cm (NEWSTAR)
3. 1.5 mL microcentrifuge tubes (Axygen, catalog number: MCT-150-C)
4. 0.2 mL microcentrifuge tubes (Axygen, catalog number: PCR-02-C)
5. 50 mL microcentrifuge tubes (Biosharp, catalog number: CT-002-50A)
6. 0.22 μm syringe filters (Merck Millipore, catalog number: SLGPR33RB)
7. Plastic spreader (Biosharp, catalog number: BS-PS-A)
8. Sterile Petri dishes (9 cm dish) (Biosharp, catalog number: BS-90-D)
9. Surgical blades (Aladdin, catalog number: C3028-02-100EA)
10. Disposable plastic cups (CHEMATE, catalog number: 0861-1119)
11. Vermiculite (Santaibio, catalog number: ZS-02-04)
12. Nutrient soil (Scotts Miracle-Gro, universal type)
13. 250 mL Erlenmeyer flask (PYREX, catalog number: 4980-250)
Equipment
1. Microvolume spectrophotometer (IMPLEN, model: NanoPhotometer® N50)
2. Growth chamber (HilPoint, model: FH-600)
3. Centrifuge (HERMLE Labortechnik GmbH, model: Z216MK)
4. Temperature-controlled shaking incubator (XiangShang, model: XS-ZD3)
5. Biochemical incubator (BLabotery, model: SPL-80)
6. Ultra-low temperature freezer (Sonyo Refrigeration, model: MDF-682)
7. Autoclave (Yamato, model: SQ510C)
8. Gel imaging system (Shenhua Science Technology, model: SH-520)
9. PCR thermal cycler (Bio-Rad Laboratories, model: T100)
10. Nucleic acid electrophoresis system (Beijing Junyi-Dongfang Electrophoresis Equipment, model: JY600E)
11. Chemical fume hood (WuXi Jinhongyang Technology, model: BM220)
Procedure
文章信息
稿件历史记录
提交日期: Dec 30, 2025
接收日期: Feb 11, 2026
在线发布日期: Feb 27, 2026
出版日期: Mar 20, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
如何引用
Zhang, Z., Wang, Q., Geng, Y. and Zhao, J. (2026). A Rapid and Visual Soybean Hairy Root Transformation Protocol Using the RUBY Reporter. Bio-protocol 16(6): e5635. DOI: 10.21769/BioProtoc.5635.
分类
植物科学 > 植物转化 > 农杆菌介导的转化方法
分子生物学
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link