- EN - English
- CN - 中文
A Rapid and High-Recovery Extracellular Vesicle (EVs) Isolation Technique from Blood Samples
一种快速高回收率的血液样本细胞外囊泡分离方法
发布: 2026年03月05日第16卷第5期 DOI: 10.21769/BioProtoc.5632 浏览次数: 23
评审: Alessandro DidonnaAnonymous reviewer(s)
Abstract
Extracellular vesicles (EVs) circulating in blood serve as non-invasive “liquid biopsies,” carrying molecular cargo that reflects the physiological and pathological state of distant cells. Their analysis is crucial for understanding disease mechanisms and discovering novel biomarkers. Clinically, blood EVs hold significant promise for early disease diagnosis, prognostic assessment, and monitoring treatment response in diverse areas such as organ transplantation, cancer, and neurological disorders. Current EV isolation techniques, beyond ultracentrifugation, include size exclusion chromatography (separation by size for high purity) and immunoaffinity capture (using antibodies for high specificity). Here, we present a simplified, rapid, and reproducible method for isolating EVs from small-volume blood samples. This protocol consistently yields a concentrated EV pellet covering 50–300 nm EVs, amenable to direct downstream analysis. Developed and validated in our laboratory using human, porcine, and murine blood samples, this method has proven instrumental in identifying EV-based biomarkers for predicting outcomes related to organ transplantation. The protocol’s adaptability and reliance on readily prepared, cost-effective reagents further enhance its utility. This scalable approach can be further integrated with subsequent purification or enrichment steps to optimize sample preparation for protein and nucleic acid assays.
Key features
• This method uses Ficoll-isolated plasma or centrifuged serum from any source, fresh to frozen.
• Protocol is easy to handle, fast, and has fewer technical details.
• 50–200 μL of plasma can be used to isolate enough EVs to run 5–8 western blot gels.
• Protocol acquires 50–300 nm EVs enriched with markers and nucleic acids.
• EVs isolated by this method can be further purified.
Keywords: EVs isolation (细胞外囊泡分离)Graphical overview
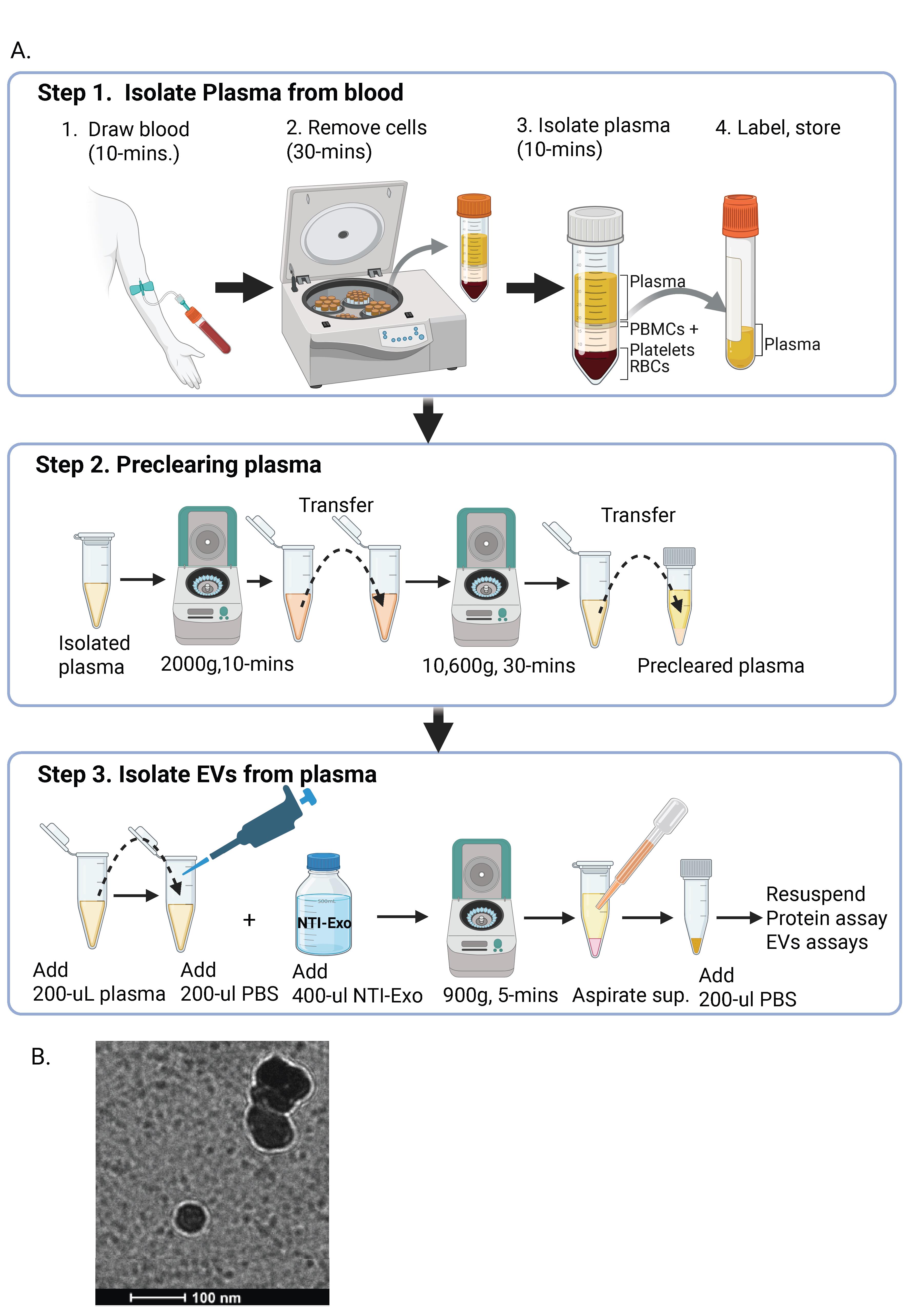
Graphical overview of extracellular vesicle (EV) isolation from plasma samples. (A) Flowchart of the isolation of extracellular vesicles from plasma, detailing plasma preparation and EV purification. (B) Transmission electron microscopy (TEM) image of EVs isolated from human plasma samples. Scale bar, 100 nm.
Background
Extracellular vesicles (EVs), once considered mere cellular debris, have emerged as critical mediators of intercellular communication, playing pivotal roles in both physiological processes and pathological conditions. These diverse nanoscale membrane-bound structures, including exosomes and microvesicles, are secreted by virtually all cell types and carry a rich cargo of proteins, lipids, and nucleic acids that reflect the state of their parent cells. This intrinsic biological function has positioned EVs as promising biomarkers for disease diagnosis, monitoring treatment efficacy, and predicting disease progression across a wide spectrum of medical conditions [1]. The rapid growth in EV research has greatly advanced our understanding of their biological role and potential clinical use. The ability to non-invasively sample and analyze patient-derived EVs offers an unparalleled opportunity for personalized medicine, moving beyond bulk tissue analysis to provide real-time insights into disease dynamics [2]. For instance, the presence of specific EV-encapsulated proteins or nucleic acids can serve as early indicators of disease onset, help to track the effectiveness of therapeutic interventions, or even predict patient response to particular treatments. This diagnostic and prognostic potential has fueled an intense drive to integrate EV analysis into routine clinical practice [3]. However, despite their immense promise, the widespread clinical application and comprehensive exploration of EVs face a significant bottleneck: the limitations of current isolation techniques. The inherent complexity and heterogeneity of biological fluids, coupled with the nanoscale dimensions of EVs, present formidable challenges. Several methods have traditionally been employed for EV isolation, each with its own set of advantages and disadvantages [4,5]. Ultracentrifugation (UC), long considered the gold standard, relies on differential centrifugation steps to progressively pellet EVs based on their size and density. While effective for isolating a broad range of EVs, UC is often criticized for its time-consuming nature, requirement for specialized equipment, potential for EV damage due to high G-forces, and the co-precipitation of contaminating proteins and aggregates, all of which necessitate skilled operators. Another common approach involves the use of size exclusion chromatography (SEC), which separates EVs based on their hydrodynamic radius as they pass through a porous matrix. SEC generally yields higher purity EV preparations and causes less mechanical stress compared to UC. However, SEC can suffer from lower recovery rates, particularly for smaller EVs, and can also be time-intensive, demanding careful column preparation and maintenance. Finally, commercial precipitation reagents offer a more convenient, often kit-based, approach to EV isolation. These methods typically involve the use of polymers that alter the solubility of EVs, causing them to aggregate and precipitate out of solution. While user-friendly and relatively rapid, precipitation methods can be prone to co-precipitation of non-EV components, leading to lower purity and potentially affecting downstream analyses. Furthermore, these reagents can sometimes interfere with the integrity or functionality of the isolated EVs.
Across all these established methods, recurring challenges persist for inconsistencies in yield and purity, compromises in EV integrity, and significant variations in reproducibility between laboratories. These issues are exacerbated when dealing with clinical samples of limited availability, such as those from pediatric patients, or when frequent, serial sampling is required for disease monitoring. The need for larger sample volumes, coupled with the labor-intensive nature of current techniques, often precludes their application in settings where only a few drops of blood are obtainable or where rapid turnaround times are crucial. The critical need for a more efficient, reliable, and accessible EV isolation method is underscored by the examples of disease-specific exosomal marker detection. Our laboratory’s investigations, including the identification of loss of LKB1 in transplant patients, SARS-CoV-2 spike protein in COVID-19 patients, and PD-L1 monitoring in immunotherapy recipients, all underscore the critical need for sensitive and accurate EV isolation from patient samples [6,7]. The lack of a robust, high-throughput, and low-volume compatible isolation technique has historically limited the potential for these exciting diagnostic and prognostic applications to translate from research laboratories into routine clinical practice.
This protocol is based on the understanding that an average EV content in human plasma is approximately 1010 per mL (ranging from 108 to 1013 per mL) [8,9]. This method, which has been validated and is routinely used, directly tackles persistent limitations by offering a rapid, user-friendly, high-recovery EV isolation technique for accessible clinical samples like blood [6]. Such an advancement is not merely incremental; it is fundamental to unlocking the full potential of EV-centric diagnostics, monitoring, and therapeutic strategies, ultimately propelling the frontiers of biomedical research and its application in personalized medicine.
Materials and reagents
Reagents
1. Commercial human serum (Sigma-Aldrich, catalog number: H4522)
2. Bovine serum albumin (BSA) (Sigma-Aldrich, catalog number: A886)
3. Dithiothreitol (DTT) (Sigma-Aldrich, catalog number: 3860-OP), store at -20 °C
4. Trizma base (Sigma-Aldrich, catalog number: RDD008)
5. EDTA (Sigma-Aldrich, catalog number: E6758)
6. Sodium chloride (NaCl) (Sigma-Aldrich, catalog number: S9888)
7. PEG (Sigma, catalog number: 81260)
8. Plasma [mice (C57BL/6J mice), or porcine (Yorkshire)], store at -20 °C
9. Anticoagulant: heparin (Sigma, catalog number: H3149)
10. 70% ethanol (for site disinfection, cotton-tipped applicators, and surgical gloves)
11. PierceTM BCA Protein Assay Kit (Thermo Scientific, catalog number: 23227)
12. RIPA lysis buffer
13. Sodium chloride (Sigma, catalog number: S5886)
14 Trizma base (Sigma, catalog number: 93352)
15. Deionized water (Milli-Q water)
16. Phosphate-buffered saline (PBS) (Sigma-Aldrich, catalog number: P4417)
17. Sodium acetate (JD Baker, catalog number:81260)
Primary antibodies used to identify EV markers
1. CD9 (BioLegend, catalog number: 312102)
2. CD63 (ProteinTech catalog number: 25682-1)
3. CD80 (ProteinTech, catalog number: 66406-1)
4. CD81 (Cell Signaling, catalog number: 56039)
5. Flotulin-1 (BioOrbit, catalog number: orb18698)
6. proteasome 20 s (Santa Cruz, catalog number: sc-271187)
7. NFKB (ProteinTech, catalog number: 66535)
8. Alix (Abcam, catalog number: ab88388)
9. TSG101 (Abcam catalog number: ab83)
10. CD63 (Santa Cruz Biotechnology, catalog number: sc-365604)
11. PD-L1 (Abcam, catalog number: ab213480)
12. CD73 (Cell Signaling Technology, catalog number: D7F9A)
13. ApoA1 (Cell Signaling, catalog number: 3350S)
14. Albumin (Cell Signaling, catalog number: 4929S)
15. Kappa light chain HRP (Thermo Fisher Scientific, catalog number: A18853)
Solutions
1. NTI-EXO solution (see Recipes)
Recipes
1. NTI-EXO solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Polyethylene glycol | 22.5% | 22.5 g |
| EDTA | 10 mM | 0.292 g |
| NaCl | 200 mM | 1.17 g |
| Tris-Cl | 200 mM | 2.42 g |
| Total | 100 mL |
Adjust to pH 7.2.
Laboratory supplies
1. 96-well flat-bottom plate (Thermo Scientific, catalog number: 21-377-832)
2. 1.5 mL Eppendorf tube (Fisher Scientific, catalog number: 50-274-4907)
3. Blood collection tubes (Fisher Scientific, catalog number: 22-040-134)
4. Cryovials (Simport Scientific, catalog number: T311-2)
5. 20 mL Syringe (BD, catalog number: 1625)
6. 0.2 µm sterile syringe filter (Thermo Scientific, catalog number: 723-2520)
7. Collection gear: 1 or 3 mL syringe with a 23–25 gauge needle
8. Polycarbonate ultracentrifuge tubes, 3 mL (Beckman Coulter, catalog number: 355635)
9. Surgical instruments: sharp dissecting scissors and thumb forceps
10. Graduated cylinder
11. Volumetric flasks
12. Sterile storage bottles
Equipment
1. Ultracentrifuge (Beckman Coulter, model: Optima XE-90) with a 70.1 TI fixed-angle titanium rotor
2. Nanosight NS300 (Software, Nanosight, NTA 3.3)
3. ExoView R200 platform (Software, ExoViewer 2.5.0)
4. pH meter (Thermo Scientific, model: Accumet AE150)
Procedure
文章信息
稿件历史记录
提交日期: Dec 29, 2025
接收日期: Feb 8, 2026
在线发布日期: Feb 24, 2026
出版日期: Mar 5, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
如何引用
Sankpal, A. N. and Sankpal, N. V. (2026). A Rapid and High-Recovery Extracellular Vesicle (EVs) Isolation Technique from Blood Samples. Bio-protocol 16(5): e5632. DOI: 10.21769/BioProtoc.5632.
分类
医学
分子生物学
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link