- EN - English
- CN - 中文
Development, Expansion, and Histological Characterization of Patient-Derived Liver Organoids for Drug Screening and Disease Modeling
用于药物筛选与疾病建模的患者来源肝脏类器官的建立、扩增及组织学特征分析
发布: 2026年03月05日第16卷第5期 DOI: 10.21769/BioProtoc.5631 浏览次数: 34
评审: Hsih-Yin TanAnonymous reviewer(s)
Abstract
Organoids are self-organizing 3D tissues representing an innovative technology with interesting implications and potential for the study of tumor biology. They can be developed from fine-needle biopsies or resection material from healthy or tumor tissues. Patient-derived organoids are able to retain most of the histological characteristics, the expression profile, and the genomic landscape of the corresponding primary tissues, making them suitable for translational studies and for the identification of molecular alterations in the field of personalized medicine. Here, we describe a detailed protocol for the preparation and in vitro expansion of tumor and non-tumor organoids from surgical resections or needle biopsies of patients with hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (iCCA), enabling subsequent testing of small-molecule VDAC1 antagonists at different doses. In parallel, we developed a hepatic steatosis model by treating healthy liver organoids with oleic acid, recapitulating key features of lipid accumulation and metabolic dysfunction in vitro. This protocol enables the generation of patient-derived liver organoids that preserve the histological and molecular characteristics of their original tissue, providing a robust and versatile platform for translational studies, personalized drug testing, and the exploration of novel therapeutic strategies targeting tumor metabolism.
Key features
• Enables reliable establishment of liver organoids from small patient samples, including fine-needle biopsies, suitable for both tumoral and non-tumoral tissue.
• Supports the generation of disease-relevant metabolic models, such as hepatic steatosis, enabling the controlled in vitro recapitulation of lipid accumulation and metabolic dysfunction.
• Standardized and reproducible platform enabling longitudinal investigation of tumor biology and therapeutic response.
• Adaptable system supporting drug screening and co-culture with stromal or immune cell components.
Keywords: Liver organoids (肝类器官)Graphical overview
Background
Liver diseases, including hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (iCCA), pose significant global health challenges and contribute substantially to cancer-related mortality. These malignancies display marked molecular heterogeneity, and reliable human models for their investigation remain limited [1]. Conventional two-dimensional cell lines do not accurately replicate the complexity of liver architecture, cellular interactions, or patient-specific genetic variability; while animal models offer valuable insights, interspecies differences limit the translational applicability of these findings to human disease [2].
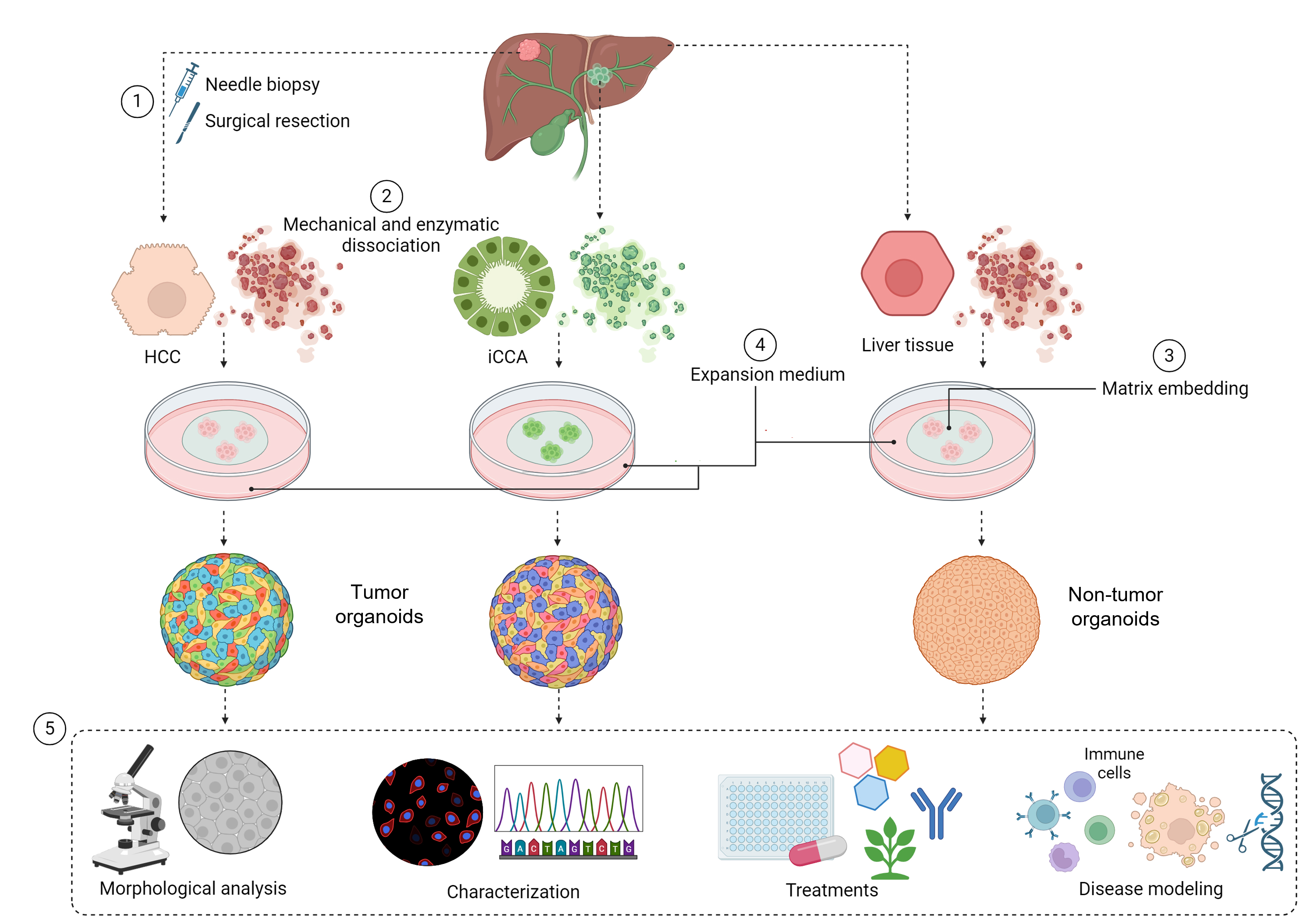
Three-dimensional (3D) organoid cultures provide a powerful way to overcome many of the limitations of traditional models. These self-organizing tissue structures can be grown from fine-needle biopsies or surgical samples taken from both tumor and healthy liver tissue. Because patient-derived organoids retain much of the architecture and gene expression patterns of the original tissue, they offer an opportunity to study disease in a way that is truly personalized. They are useful not only for modeling liver disorders and exploring the underlying biology but also for testing drugs in conditions that closely reflect the patient’s own tissue environment. Another advantage is their durability: organoids can be expanded over long periods in culture and cryopreserved, making them a flexible and accessible resource for ongoing and future research [3,4].
Although protocols for liver and primary liver cancer organoid cultures have been previously reported, variations in tissue sources, processing workflows, and downstream applications often limit standardization and reproducibility across laboratories. This protocol was developed to provide a standardized and reproducible workflow for the development, long-term maintenance, and functional use of patient-derived liver organoids, with a success rate of 28% for HCC, 76% for iCCA, and 91% for normal liver tissue. The lower establishment efficiency observed for HCC-derived organoids reflects intrinsic biological characteristics of hepatocyte-derived tumors and their strong dependence on tumor differentiation and stage [5].
Organoids are generated from both tumor and non-tumor tissue, starting from surgical resections or needle biopsies, enabling their application in disease modeling, drug screening, and identification of novel therapeutic targets. The method includes processing the tissue, starting the culture, and maintaining it long-term while keeping the original tissue characteristics. The protocol outlines two main uses. First, it provides a method to evaluate small molecules that modulate mitochondrial metabolism, such as VDAC1 antagonists in tumor organoids. VDAC1 is a mitochondrial outer membrane ion channel that is highly expressed in glycolytic tumor cells and contains a putative binding pocket for small molecules. Binding of these compounds can displace NADH, induce mitochondrial stress, alter outer mitochondrial membrane permeability, and promote apoptotic signaling [6]. Second, it offers a model for fatty liver disease by treating healthy liver organoids with oleic acid, mimicking key features of hepatic steatosis, including lipid accumulation and metabolic dysfunction. Together, these applications highlight the versatility of patient-derived liver organoids as a research tool. The protocol provides a robust, physiologically relevant model for studying liver tumor biology, drug responses, and metabolic regulation, bridging basic and translational research and supporting the development of targeted therapies for liver cancers and metabolic diseases.
Materials and reagents
Biological materials
1. Patient-derived resections and fine-needle biopsies (Fondazione IRCCS Policlinico San Matteo, Pavia, Italy)
The study was conducted in accordance with the Declaration of Helsinki (2013) and the Declaration of Istanbul (2018) and was approved by the Ethics Committee of the Fondazione IRCCS Policlinico San Matteo, Pavia (Prot. 20200020546, Prot. 0008508/24, and NCT06899854).
Reagents
Antibodies
1. Albumin polyclonal antibody, BSA-free (Novus Biologicals, catalog number: NBP2-38174)
2. Alpha-Feto protein, AFP polyclonal antibody (Invitrogen, catalog number: PA5-21004)
3. CD326 (EpCAM) monoclonal antibody, Alexa FluorTM (clone MH99) (Invitrogen, catalog number: 53-8326-42)
4. Cytokeratin-7 monoclonal antibody (clone OV-TL12/30) (Novus Biologicals, catalog number: NBP2-44814)
5. Cytokeratin-19 monoclonal antibody (clone ROCK108) (Invitrogen, catalog number: MA1-06329)
6. Ki67 monoclonal antibody (clone SP6) (Invitrogen, catalog number: MA5-14520)
7. Goat anti-mouse IgG (H + L) secondary antibody, Alexa FluorTM 488 (Invitrogen, catalog number: A-11001)
8. Goat anti-rabbit IgG (H + L) secondary antibody, Alexa FluorTM 647 (Invitrogen, catalog number: A-21244)
Culture media, supplements, and growth factors
1. A83-01 (Sigma-Aldrich, catalog number: SML0788)
2. Advanced DMEM/F-12 (AdDMEM/F-12) (Gibco, catalog number: 3700-100-01)
3. B-27 supplement, without vitamin A (Gibco, catalog number: 12587010)
4. BMP-7, recombinant human (Peprotech, catalog number: 120-03P)
5. DAPT (Peprotech, catalog number: 2088055)
6. Dexamethasone (Sigma-Aldrich, catalog number: D4902)
7. DMEM high glucose with L-glutamine and sodium pyruvate (EuroClone, catalog number: ECM0728)
8. Epidermal growth factor (EGF), recombinant human (R&D Systems, catalog number: 236-EG-200)
9. Fetal bovine serum (FBS) (Gibco, catalog number: 10270-106)
10. FGF-19, recombinant human (Peprotech, catalog number: 100-32)
11. Forskolin (Tocris, catalog number: 1099/10)
12. Gastrin I human (Leu15) (Sigma-Aldrich, catalog number: G9145)
13. GlutaMAXTM supplement (Gibco, catalog number: 51985-026)
14. Hepatocyte growth factor/scatter factor (HGF/SF) (homemade) [7]
15. L-WRN conditioned medium (Sigma-Aldrich, catalog number: SCM105)
16. MEM non-essential amino acids 100× w/o L-glutamine (Biowest, catalog number: X0557)
17. N-2 supplement (Gibco, catalog number: 17502048)
18. N-acetyl-L-cysteine (Sigma-Aldrich, catalog number: A9165)
19. Nicotinamide (Sigma-Aldrich, catalog number: N0636)
20. Opti-MEMTM reduced serum medium (Gibco, catalog number: 31985062)
21. Penicillin–streptomycin (Gibco, catalog number: 15140-122)
22. Recovery cell culture freezing medium (Gibco, catalog number: 12648010)
23. RPMI 1640 medium (EuroClone, catalog number: ECB9006)
24. R-Spondin-1 conditioned medium (Sigma-Aldrich, catalog number: SCM104)
25. Y-27632 dihydrochloride (ROCK inhibitor) (Tocris, catalog number: 1254/1)
Buffers, staining reagents, and general chemicals
1. ACK lysing buffer (Gibco, catalog number: A1049201)
2. Acetone (Sigma-Aldrich, catalog number: 179124)
3. Antibiotic–antimycotic solution (Sigma-Aldrich, catalog number: A5955)
4. BODIPYTM 493/503 (Invitrogen, catalog number: D3922)
5. Bovine serum albumin (BSA) (Pan-Biotech, catalog number: P06-1391500)
6. BSA, fatty acid free (Sigma-Aldrich, catalog number: A1595)
7. CellTiter-Glo 3D cell viability assay (Promega, catalog number: G9681)
8. Cultrex organoid harvesting solution (R&D Systems, catalog number: BME001-10)
9. Cultrex UltiMatrix reduced growth factor basement membrane extract (R&D Systems, catalog number: BME001-10)
10. Diaminobenzidine tetrahydrochloride (DAB) (Biocare Medical, catalog number: DB801)
11. Diva Decloaker solution 100× (Bio-Optica, catalog number: BRI2006L)
12. DMSO (Sigma-Aldrich, catalog number: 472301)
13. Dulbecco’s phosphate-buffered saline (DPBS) (Gibco, catalog number: 14190-094)
14. Eosin (Sigma-Aldrich, catalog number: 230251)
15. Eukitt (Kindler, catalog number: D-79110)
16. Hematoxylin solution (Histo-Line Laboratories, catalog number: 01HEMCA1000)
17. HEPES buffer (Gibco, catalog number: 15630056)
18. Hoechst 33342 (Invitrogen, catalog number: H1399)
19. ImmPACTTM DAB (Vector Laboratories, catalog number: NC9567138)
20. MACS® tissue storage solution (Miltenyi Biotech, catalog number: 130-100-008)
21. Oleic acid (Sigma-Aldrich, catalog number: O1383)
22. Paraformaldehyde (PFA) (Sigma-Aldrich, catalog number: 158127)
23. Paraplast X-TRA (Sigma-Aldrich, catalog number: P3808)
24. Peroxidase-blocking solution (Dako, catalog number: S2023)
25. SlowFadeTM diamond antifade mountant (Invitrogen, catalog number: S36967)
26. Trypan blue stain 0.4% (Logos Biosystems, catalog number: T13001)
27. Triton X-100 (Sigma-Aldrich, catalog number: T8787)
28. Tween 20 (PanReac AppliChem, catalog number: A4974)
29. Tumor Dissociation kit, human (Miltenyi Biotech, catalog number: 130-095-929)
30. Xylene (Bio-Optica, catalog number: 06-1304F)
Solutions
1. Complete medium (see Recipes)
2. Basal medium (see Recipes)
3. Expansion medium (see Recipes)
4. Differentiation medium (see Recipes)
5. Opti-MEM Medium (see Recipes)
6. Oleic acid/BSA reagents (see Recipes)
Recipes
1. Complete medium
| Reagent | Stock concentration | Volume |
|---|---|---|
| DMEM high glucose w/ L-glutamine w/ sodium pyruvate | 1× | 88 mL |
| Fetal Bovine Serum (FBS) | - | 10 mL |
| Antibiotic Antimycotic Solution | 100× | 1 mL |
| MEM non-essential amino acids | 100× | 1 mL |
| Total | 100 mL |
2. Basal medium
| Reagent | Stock concentration | Volume |
|---|---|---|
| AdDMEM/F-12 | 1× | 485 mL |
| Pen/Strep | 100× | 5 mL |
| GlutaMAX | 100× | 5 mL |
| HEPES | 1 M | 5 mL |
| Total | 500 mL |
3. Expansion medium
| Reagent | Stock concentration | Weight | Diluent | Solubility | Volume | Aliquot storage |
|---|---|---|---|---|---|---|
| Basal medium | 1× | 5,552 mL | ||||
| B27 | 50× | - | - | - | 200 μL | -20 °C |
| N2 | 100× | - | - | - | 100 μL | -20 °C |
| Nicotinamide | 1 M | 611 mg | 5 mL | DPBS | 100 μL | -20 °C |
| NAC | 500 mM | 408 mg | 5 mL | 37 °C H2O | 25 μL | -20 °C |
| Y-27632 | 100 mM | 10 mg | 0.308 mL | H2O | 1 μL | -20 °C |
| Forskolin | 10 mM | 10 mg | 2.44 mL | DMSO | 10 μL | -20 °C |
| A83-01 | 5 mM | 5 mg | 2.37 mL | DMSO | 10 μL | -20 °C |
| [Leu15]-gastrin I | 200 μM | 1 mg | 2.40 mL | DPBS | 0.5 μL | -20 °C |
| EGF | 500 μg/mL | 200 μg | 0.400 mL | DPBS | 1 μL | -20 °C |
| HGF/SF | 0.35 mg/mL | 0.7 μL | -20 °C | |||
| R-Spondin1 | - | - | - | - | 1 mL | -20 °C |
| Wnt3a | - | - | - | - | 3 mL | -20 °C |
| Total | 10 mL |
The expansion medium is composed of basal medium with the addition of B27 (1:50), N2 (1:100), 10 mM nicotinamide, 1.25 mM N-acetyl-L-cysteine (NAC), 10 μM forskolin, 10 μM Y-27632 dihydrochloride (ROCK inhibitor), 5 μM A83-01, 10 nM [Leu15]-gastrin I, 50 ng/mL epidermal growth factor (EGF), 25 ng/mL hepatocyte growth factor/scatter factor (HGF/SF) (homemade), 10% R-Spondin1 conditioned medium, and 30% Wnt3a conditioned medium with Noggin.
4. Differentiation medium
| Reagent | Stock concentration | Weight | Diluent | Solubility | Volume | Aliquot storage |
|---|---|---|---|---|---|---|
| Basal medium | 1× | 9,517 mL | ||||
| B27 | 50× | - | - | - | 200 μL | -20 °C |
| N2 | 100× | - | - | - | 100 μL | -20 °C |
| A83-01 | 5 mM | 5 mg | 2.37 mL | DMSO | 1 μL | -20 °C |
| [Leu15]-gastrin I | 200 μM | 1 mg | 2.40 mL | DPBS | 0.5 μL | -20 °C |
| EGF | 500 μg/mL | 200 μg | 0.400 mL | DPBS | 1 μL | -20 °C |
| HGF/SF | 0.35 mg/mL | De Nola et al. [7] | 0.7 μL | -20 °C | ||
| FGF-19 | 100 μg/mL | 5 μg | 50 μL | DPBS | 10 μL | -20 °C |
| DAPT | 10 mM | 10 mg | 2.31 mL | DMSO | 10 μL | -20 °C |
| BMP-7 | 25 μg/mL | 2 μg | 80 μL | DPBS | 10 μL | -20 °C |
| Dexamethasone | 2 mM | 25 mg | 32 mL | EtOH | 150 μL | -20 °C |
| Total | 10 mL |
The differentiation medium is composed of basal medium with the addition of B27 (1:50), N2 (1:100), 500 nM A83-01, 10 nM [Leu15]-gastrin I, 50 ng/mL EGF, 25 ng/mL HGF/SF (homemade), 100 ng/mL fibroblast growth factor 19 (FGF-19), 25 ng/mL BMP-7, 10 μM DAPT, and 30 μM dexamethasone.
5. Opti-MEM medium
| Reagent | Stock concentration | Weight | Diluent | Solubility | Volume | Aliquot storage |
|---|---|---|---|---|---|---|
| Opti-MEM | 1× | 4.73 mL | ||||
| Pen/Strep | 100× | 50 μL | -20 °C | |||
| B27 | 50× | - | - | - | 100 μL | -20 °C |
| N2 | 100× | - | - | - | 50 μL | -20 °C |
| Nicotinamide | 1 M | 611 mg | 5 mL | DPBS | 50 μL | -20 °C |
| NAC | 500 mM | 408 mg | 5 mL | 37 °C H2O | 12.5 μL | -20 °C |
| Y-27632 | 100 mM | 10 mg | 0.308 mL | H2O | 0.5 μL | -20 °C |
| Forskolin | 10 mM | 10 mg | 2.44 mL | DMSO | 5 μL | -20 °C |
| A83-01 | 5 mM | 5 mg | 2.37 mL | DMSO | 5 μL | -20 °C |
| [Leu15]-gastrin I | 200 μM | 1 mg | 2.40 mL | DPBS | 0.25 μL | -20 °C |
| EGF | 500 μg/mL | 200 μg | 0.400 mL | DPBS | 0.5 μL | -20 °C |
| HGF/SF | 0.35 mg/mL | 0.35 μL | -20 °C | |||
| Total | 5 mL |
The medium is composed of Opti-MEM with 1% penicillin/streptomycin, supplemented with B27 (1:50), N2 (1:100), 10 mM nicotinamide, 1.25 mM N-acetyl-L-cysteine (NAC), 10 μM forskolin, 10 μM Y-27632, 5 μM A83-01, 10 nM [Leu15]-gastrin I, 50 ng/mL EGF, and 25 ng/mL HGF/SF.
6. Oleic acid/BSA reagents
| Stock concentration | Reagent 1 | Volume 1 | Diluent 2 | Volume 2 | Additional information |
|---|---|---|---|---|---|
| 1 M oleic acid | 3.15 M oleic acid | 1.12 mL | 100% ethanol | 3.5 mL | |
| 37.5 mM NaOH | 100 mM NaOH | 1.5 mL | Sterile H2O | 2.5 mL | |
| 20 mM sodium oleate | 1 M oleic acid | 80 μL | 37.5 Mm NaOH | 3,920 μL | Vortex and heat at 70 °C for 30 min |
| 5% BSA/DPBS solution | 30% BSA fatty acid free | 2 mL | DPBS | 10 mL | |
| 5 mM sodium oleate | 20 mM sodium oleate | 4 mL | DPBS + 5% BSA/DPBS | 12 mL | Vortex and heat at 37 °C for 1 h |
| CTRL- solution | 37.5 mM NaOH | 4 mL | 5% BSA/DPBS | 12 mL | Heat at 37 °C for 1 h |
All solutions are aliquoted and stored at -20 °C.
Laboratory supplies
1. 10 μL pipette tips (Clearline, catalog number: 713130H)
2. 20 μL pipette tips (Clearline, catalog number: 014210CLH)
3. 200 μL pipette tips (Clearline, catalog number: 713117H)
4. 1,250 μL pipette tips (Clearline, catalog number: 713116H)
5. 10 mL serological pipettes (Greiner Bio-One, catalog number: 607180)
6. 15 mL tubes (Falcon, catalog number: 10773501)
7. 2 mL cryovials (Clearline, catalog number: CL2ARBEPSTS)
8. 25 mL serological pipettes (Greiner Bio-One, catalog number: 760160-TRI)
9. 50 mL tubes (Falcon, catalog number: 10788561)
10. Cell culture multi-well plates (24-well) (Nunc, catalog number: 144530)
11. Cell culture multi-well plates (48-well) (Nunc, catalog number: 150787)
12. gentleMACSTM C tubes (Miltenyi Biotech, catalog number: 130-093-237)
13. Ibidi chamber slides (ibidi GmbH, catalog number: 80841)
14. MACS® SmartStrainers (70 μm) (Miltenyi Biotech, catalog number: 130-098-462)
15. Petri dishes (Greiner Bio-One, catalog number: 760160-TRI)
Equipment
1. Automated cell counter (Luna, catalog number: L40002)
2. Cell culture incubator (Thermo Fisher Scientific, model: MSC Advantage 1.2)
3. Confocal microscope (Leica, model: TCS SP5)
4. GentleMACSTM dissociator (Miltenyi Biotech, catalog number: 130-093-235)
5. Light microscope (Nikon, model: Eclipse Ts2-FL Inverted)
6. Microfuge 20R (Beckman Coulter, catalog number: B30150)
7. Microtome (Leica, model: RM2155)
8. MSC Advantage 1.2 Class II biological safety cabinet (Thermo Fisher Scientific, catalog number: 51025411)
Software and datasets
1. ImageJ 1.53c (NIH, https://imagej.net/ij/)
2. Prism (GraphPad) version 8.3.0 (Dotmatics, https://www.graphpad.com)
Procedure
文章信息
稿件历史记录
提交日期: Nov 19, 2025
接收日期: Jan 29, 2026
在线发布日期: Feb 24, 2026
出版日期: Mar 5, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
如何引用
De Siervi, S., Mantovani, S., Oliviero, B., Mondelli, M. U., Di Noia, M., Soldani, C., Franceschini, B., De Luca, F., Maestri, M., Corallo, S., Lolicato, M., Cannito, S. and Turato, C. (2026). Development, Expansion, and Histological Characterization of Patient-Derived Liver Organoids for Drug Screening and Disease Modeling. Bio-protocol 16(5): e5631. DOI: 10.21769/BioProtoc.5631.
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link