- EN - English
- CN - 中文
Selective Isolation of TOP3B•mRNA Covalent Intermediates Using Denaturing Oligo-dT Pulldown
利用变性Oligo-dT Pull-down技术选择性分离TOP3B-mRNA共价中间体
发布: 2026年03月05日第16卷第5期 DOI: 10.21769/BioProtoc.5627 浏览次数: 35
评审: Willy R Carrasquel-UrsulaezAnu ThomasAnonymous reviewer(s)

相关实验方案
基于适体的 mRNA 亲和纯化程序 (RaPID) 用于鉴定酵母中的相关 RNA (RaPID-seq) 和蛋白质 (RaPID-MS)
Rohini R. Nair [...] Jeffrey E. Gerst
2022年01月05日 5403 阅读
Abstract
The deletion and mutation of Topoisomerase 3β (TOP3B) is linked to multiple neurological disorders and is the only known topoisomerase that is also catalytically active on RNA in vitro and in cells. Uniquely, TOP3B is primarily localized to the cytoplasm, binds to open reading frames of mRNA, and regulates mRNA stability and translation in a transcript-specific manner. A common approach for studying TOP3B activity in cells is immunodetection of TOP3B•RNA covalent intermediates after bulk RNA isolation. However, in this approach, the RNA species is unknown and is not selective for the major TOP3B substrate, mRNA. In this protocol, we describe a recently developed and optimized protocol for capturing TOP3B•mRNA covalent intermediates using oligo-dT isolation of mRNA under protein-denaturing conditions. Covalent intermediates are then detected by a dual membrane slot blotting strategy with nitrocellulose and positively charged nylon membranes. Nitrocellulose membrane-bound TOP3B•mRNA covalent intermediates are analyzed by immunodetection, and nylon membrane-bound free mRNA is stained with methylene blue. The protocol detailed below has been validated with wildtype and mutant 3xFLAG-tagged TOP3B expressed in Neuro2A cells, with additional optimization for slot blotting using recombinant EGFP.
Key features
• This protocol is optimized for isolation of TOP3B•mRNA covalent intermediates from cultured mammalian cells.
• Slot blotting allows for higher throughput and sensitive detection of TOP3B•mRNA covalent intermediates and allows for free mRNA to serve as a loading control.
• Alternative to laborious extraction methods that do not select for TOP3B covalently linked to mRNA over other RNA species.
• Can be completed in two days (not including variable time for mammalian cell sample collection).
Keywords: mRNA pulldown (mRNA 拉下实验)Graphical overview
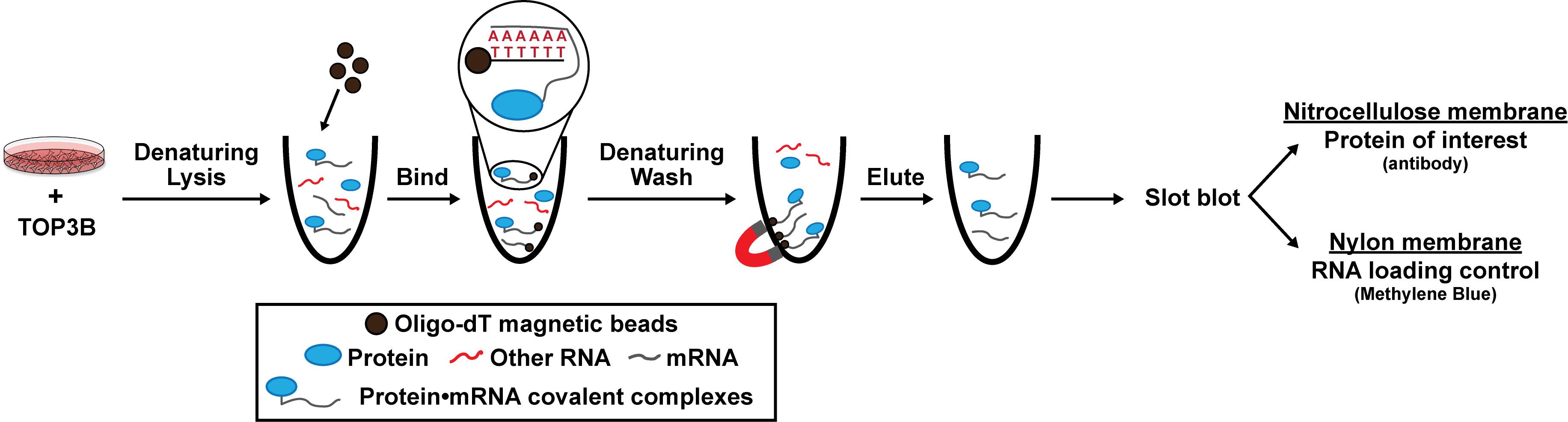
Schematic of denaturing oligo-dT isolation of TOP3B•mRNA covalent intermediates
Background
RNA-binding proteins (RBPs) have diverse functions that are critical for every level of gene expression and are linked to multiple human diseases [1,2]. Deletion and mutation of Topoisomerase 3β (TOP3B) are linked to schizophrenia, autism spectrum disorder, and intellectual disability [3–7]. Notably, TOP3B is the only human topoisomerase that functions on both DNA and RNA, and multiple reports suggest that mRNA is its primary substrate [4,8,9]. Unlike most other RBPs, but similar to other topoisomerases, TOP3B forms an intermediate that is covalently linked to the 5′ phosphate of its substrate RNAs during the catalytic cycle. CLIP-based studies have shown that TOP3B primarily binds to the open reading frame (ORF) of mRNAs [4,9], which is common for RBPs that have a role in mRNA translation and post-transcriptional gene regulation. Additionally, recent ribosome profiling data indicated that TOP3B regulates the translation of some mRNAs by acting as a traditional RBP where catalytic activity is not necessary, while other mRNAs are regulated by TOP3B in a catalytic activity-dependent manner [9]. Therefore, it is important to distinguish the mRNA-binding capability from catalytic cleavage activity of TOP3B to identify which steps in the cycle may be affected by de novo mutations present in the human population and to understand mechanisms of translational control governed by TOP3B. However, published cell-based assays are limited due to laborious cesium chloride gradients and multiple large-scale extractions from the interface of acidic guanidinium thiocyanate-phenol-chloroform-based approaches (e.g., TRIzol, TRI Reagent) and depend on assaying unidentified RNAs from cells [10–13]. As most data in the field suggest that mRNA is the primary target of TOP3B, there is a need to develop an approach to assess TOP3B activity on mRNA over other RNA species.
Here, we provide a published method to selectively evaluate covalently linked TOP3B•mRNA complexes using denaturing oligo-dT pulldown and slot blotting [14]. We ectopically express wildtype (WT) and mutant 3xFLAG-tagged TOP3B in cells via plasmid transfection and incubate the cell lysate with oligo-dT magnetic beads to selectively bind the poly(A) tail of mRNA. Lysis and wash conditions are denaturing, allowing the removal of any RBPs that are not covalently linked to mRNA and the retention of TOP3B•mRNA covalent intermediates. We then evaluate the TOP3B•mRNA intermediate levels by slot blotting for the 3xFLAG tag. As validation, we show two mutants that produce decreased and increased intermediate levels compared to the wildtype TOP3B [10,14]. Intermediate levels from a catalytically inactive TOP3B (Y336F), which can bind mRNA but cannot form covalent intermediates, are markedly reduced. Conversely, intermediate levels from a “self-trapping” mutant TOP3B (R338W), which causes accumulation of unresolved TOP3B•mRNA intermediates by preventing the rejoining step after substrate cleavage, are robustly detected. We have recently published and further validated this method using TOP3B tagged on either the N- or C-terminus, as well as with domain deletions and point mutations [14].
Materials and reagents
Biological materials
1. Neuro-2A cells (ATCC, catalog number: CCL-131)
Reagents
1. 1 M Tris-HCl, pH 7.4 (Apex Bioresearch Products, catalog number: 18-190)
2. 2 M lithium chloride (LiCl) (Sigma, catalog number: L7026-1L)
3. Lithium dodecyl sulfate (LiDS) (Sigma, catalog number: L9781-50G)
4. 500 mM EDTA, pH 8.0 (Millipore, catalog number: 324504-500mL)
5. 1 M DTT (Thermo Scientific, catalog number: R0861)
6. Oligo-dT magnetic beads (NEB, catalog number: S1419S)
7. 1% (w/v) methylene blue (Thermo Scientific, catalog number: 042771.AP)
8. 17.4 M glacial acetic acid (Fisher Chemical, catalog number: A38C-212)
9. 3 M sodium acetate, pH 5.5 (Invitrogen, catalog number: AM9740)
10. 1× phosphate-buffered saline (PBS), pH 7.4 (Quality Biological, catalog number: 114-058-131)
11. 10× Tris-buffered saline (TBS), pH 7.4 (Quality Biological, catalog number: 351-086-151)
12. 100% Tween-20 (VWR, catalog number: 0777-4L)
13. Instant non-fat dry milk (Quality Biological, catalog: A614-1001) or equivalent
14. Sodium azide (Sigma, catalog number: S2002-5G)
15. Mouse anti-FLAG M2 primary antibody (Sigma, catalog number: F3165-1mg)
16. Rabbit anti-GFP (D5.1) primary antibody (Cell Signaling Technologies, catalog number: 2956)
17. HRP-conjugated goat anti-mouse IgG (H+L) (Invitrogen, catalog number: 31430)
18. HRP-conjugated goat anti-rabbit IgG (H+L) (Invitrogen, catalog number: 31460)
19. SuperSignal West Pico PLUS chemiluminescent substrate (Invitrogen, catalog number: 34578)
20. Recombinant EGFP (Abcam, catalog number: ab134853)
21. Plasmid pCMV6/TOP3B (WT)-3xFLAG (Addgene, plasmid number: 249681)
22. Plasmid pCMV6/TOP3B (Y336F)-3xFLAG (Addgene, plasmid number: 249682)
23. Plasmid pCMV6/TOP3B (R338W)-3xFLAG (Addgene, plasmid number: 249683)
23. Ultrapure water (from Millipore ultrapure water system or equivalent)
Solutions
1. 1 M DTT (see Recipes)
2. 10% (w/v) LiDS (see Recipes)
3. Oligo-dT lysis buffer (see Recipes)
4. Oligo-dT wash I buffer (see Recipes)
5. Oligo-dT wash II buffer (see Recipes)
6. Oligo-dT low salt buffer (see Recipes)
7. Oligo-dT elution buffer (TE) (see Recipes)
8. Methylene blue staining solution (see Recipes)
9. TBST (see Recipes)
10. 5% (w/v) non-fat dry milk (see Recipes)
11. 20 mM Tris-HCl, pH 7.4 (see Recipes)
12. 2% (w/v) sodium azide (see Recipes)
13. Primary antibody solution (see Recipes)
14. Secondary antibody solution (see Recipes)
Recipes
1. 1 M DTT
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DTT | 1M | 1.55 g |
| Ultrapure water | n/a | To 10 mL |
| Total | n/a | 10 mL |
2. 10% (w/v) LiDS
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| LiDS | 10% (w/v) | 50 g |
| Ultrapure water | n/a | To 500 mL |
| Total | n/a | 500 mL |
Safety note: Wear a mask to avoid inhaling the reagent.
3. Oligo-dT lysis buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1 M Tris-HCl, pH 7.4 | 100 mM | 30 mL |
| 8 M LiCl | 500 mM | 18.75 mL |
| 10% LiDS (w/v) | 0.5% (w/v) | 15 mL |
| 500 mM EDTA, pH 8.0 | 1 mM | 600 μL |
| Ultrapure water | n/a | 235.65 mL |
| Total | n/a | 300 mL |
4. Oligo-dT wash I buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1 M Tris-HCl, pH 7.4 | 20 mM | 6 mL |
| 8 M LiCl | 500 mM | 18.75 mL |
| 10% (w/v) LiDS | 0.1% (w/v) | 3 mL |
| 500 mM EDTA, pH 8.0 | 1 mM | 600 μL |
| Ultrapure water | n/a | 271.65 mL |
| Total | n/a | 300 mL |
5. Oligo-dT wash II buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1 M Tris-HCl, pH 7.4 | 20 mM | 6 mL |
| 8 M LiCl | 500 mM | 18.75 mL |
| 500 mM EDTA, pH 8.0 | 1 mM | 600 μL |
| Ultrapure water | n/a | 274.65 mL |
| Total | n/a | 300 mL |
6. Oligo-dT low salt buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1 M Tris-HCl, pH 7.4 | 20 mM | 3 mL |
| 8 M LiCl | 200 mM | 3.75 mL |
| 500 mM EDTA, pH 8.0 | 1 mM | 300 μL |
| Ultrapure water | n/a | 142.95 mL |
| Total | n/a | 150 mL |
7. Oligo-dT elution buffer (TE)
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1 M Tris-HCl, pH 7.4 | 20 mM | 6 mL |
| 500 mM EDTA, pH 8.0 | 1 mM | 600 μL |
| Ultrapure water | n/a | 293.4 mL |
| Total | n/a | 300 mL |
8. Methylene blue staining solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1% (w/v) methylene blue | 0.2% (w/v) | 10 mL |
| 17.4 M glacial acetic acid | 0.4 M | 1.15 mL |
| 3 M sodium acetate | 0.4 M | 6.67 mL |
| Ultrapure water | n/a | 32.18 mL |
| Total | n/a | 50 mL |
Safety note: Add water first and then slowly add other reagents.
9. TBST
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 10× TBS, pH 7.4 | 1× | 100 mL |
| 100% Tween-20 | 0.1% | 1 mL |
| Ultrapure water | n/a | 900 mL |
| Total | n/a | 1 L |
10. 5% (w/v) non-fat dry milk
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Non-fat dry milk | 5% (w/v) | 10 g |
| TBST | n/a | 200 mL |
| Total | n/a | 200 mL |
11. 20 mM Tris-HCl, pH 7.4
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| 1 M Tris-HCl, pH 7.4 | 20 mM | 10 mL |
| Ultrapure water | n/a | 490 mL |
| Total | n/a | 500 mL |
12. 2% (w/v) sodium azide
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Sodium azide | 2% (w/v) | 4 g |
| Ultrapure water | n/a | To 200 mL |
| Total | n/a | 200 mL |
Safety note: Only use plastic or non-metal spatula. Wear a mask to avoid inhaling the reagent.
13. Primary antibody solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Mouse anti-FLAG M2 antibody | 1:1,000 | 10 μL |
| 2% (w/v) sodium azide | 0.2% (w/v) | 100 μL |
| TBST | n/a | 9.89 mL |
| Total | n/a | 10 mL |
14. Secondary antibody solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| HRP-conjugated goat anti-mouse IgG (H+L) | 1:30,000 | 3 μL |
| TBST | n/a | 29.997 mL |
| Total | n/a | 30 mL |
Laboratory supplies
1. 500 mL vacuum filter with 0.22 μm cellulose nitrate membrane (Corning, catalog number: 430758)
2. 0.2 μm nitrocellulose membranes (Bio-Rad, catalog number: 1620112)
3. BrightStar Plus positively charged nylon membrane (Invitrogen, catalog number: AM10104)
4. 1.7 mL microcentrifuge tubes (Olympus Plastics, catalog number: 24-282)
5. 8-strip PCR tubes (Olympus Plastics, catalog number: 27-125UA)
6. Tissue wipes (VWR, catalog number: 82003-820)
7. Paper towels
8. Plastic wrap
9. 50 mL centrifuge tubes (VWR, catalog number: 89039-656)
10. 15 mL centrifuge tubes (VWR, catalog number: 89039-664)
11. Microcentrifuge tube racks
12. Four-way tube rack
13. Blot development folders (Azure Biosystems, catalog number: AC2126)
14. 5, 10, 25, and 50 mL serological pipettes (Gen Clone, catalog numbers: 12-102, 12-104, 12-106, 12-107)
15. Glass Pasteur pipette
16. Forceps
17. p10, p200, and p1000 pipette tips (VWR, catalog numbers: 76323-388, 76323-390, 76323-456)
Equipment
1. -80 °C freezer
2. -20 °C freezer
3. 4 °C refrigerator
4. Magnetic rack (DynaMag-2) (Invitrogen, catalog number: 12321D)
5. Slot blot apparatus, Hoefer PR648 (discontinued; often available on eBay) or Bio-Dot SF microfiltration apparatus (Bio-Rad, catalog number: 1703938)
6. Three-way stopcock (Thermo Fisher, catalog number: 64700004)
7. Large plastic container (e.g., Tupperware)
8. p2.5, p10, p20, p200, p1000 pipettes
9. Western blot boxes (with lid)
10. Plastic blot container
11. Eppendorf ThermoMixer F1.5 (Eppendorf, catalog number: 5384000020) or equivalent
12. Eppendorf Centrifuge 5430 (Eppendorf, catalog number: 022620584) or equivalent
13. Benchmark Orbi-Blotter orbital shaker (Benchmark, catalog number: BT30) or equivalent
14. Benchmark BenchRocker 2D (Benchmark, catalog number: BR2000) or equivalent
15. Stratalinker UV Crosslinker 1800 (Stratagene)
16. GelDoc Go Imaging System (Bio-Rad, catalog number: 12009077) or equivalent
17. T100 thermal cycler (Bio-Rad, catalog number: 1861096) or equivalent
18. Azure Sapphire Biomolecular Imager (Azure Biosystems, catalog number: IS4000)
19. Aspirator
20. Millipore ultrapure water system
21. NanoDrop One Microvolume UV-Vis spectrophotometer (Thermo Fisher, catalog number: ND-ONE-W)
22. Benchmark Vortex BenchMixer (Benchmark, catalog number: BV1000) or equivalent
23. Mini centrifuge MLX-106-GS (Genesee Scientific, catalog number: 3377846, 3377848) or equivalent
24. Benchmark Roto-Mini Plus Rotator, variable speed (Benchmark, catalog number: R2024) or equivalent
25. Blotting roller (Thermo Fisher, catalog number: LC2100)
26. 1 L plastic beaker
27. p200 multichannel pipette
Software and datasets
1. ImageJ (https://imagej.net/ij/, free download from the NIH)
Procedure
文章信息
稿件历史记录
提交日期: Dec 3, 2025
接收日期: Jan 30, 2026
在线发布日期: Feb 10, 2026
出版日期: Mar 5, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/).
如何引用
Warrick, J. E. and Kearse, M. G. (2026). Selective Isolation of TOP3B•mRNA Covalent Intermediates Using Denaturing Oligo-dT Pulldown. Bio-protocol 16(5): e5627. DOI: 10.21769/BioProtoc.5627.
分类
医学
分子生物学 > RNA > RNA-蛋白质相互作用
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link