- EN - English
- CN - 中文
A Standardized Culture Medium for Comparative Drug Efficacy Evaluation Across Plasmodium and Babesia Species
用于疟原虫与巴贝虫药效比较评价的标准化培养基体系
发布: 2026年03月05日第16卷第5期 DOI: 10.21769/BioProtoc.5625 浏览次数: 17
评审: Prudhvi Chand MallepaddiAnonymous reviewer(s)
Abstract
The discovery of broad-spectrum antiparasitic agents relies on the ability to evaluate drug efficacy under harmonized in vitro conditions across related species. However, current drug screening pipelines for intraerythrocytic parasites are constrained by the use of species-specific media with distinct nutrient compositions and serum sources, which hinder direct comparison of compound potency. To address this gap, we describe a unified erythrocytic culture system based on DMEM/F12 supplemented with 20% fetal bovine serum (DFS20), which supports robust asexual growth of multiple Plasmodium falciparum strains (3D7, Dd2, HB3, V1/S), Babesia duncani, Babesia divergens (Rouen 87), and Babesia MO1. Parasite proliferation and morphology in DFS20 are comparable to those observed in established species-specific media such as RPMI-1640 for P. falciparum and B. divergens and HL-1/Claycomb/DMEM/F12/SFM for B. duncani, while eliminating reliance on undefined or discontinued proprietary components. Importantly, this standardized medium enables cross-species growth inhibition assays for direct comparison of drug efficacy under identical conditions. Using this platform, we recently screened dihydrotriazines and biguanides targeting the conserved DHFR-TS enzymes and identified potent antifolate candidates with broad-spectrum activity against Babesia and Plasmodium species. For B. duncani, which is uniquely supported by both a continuous in vitro human erythrocyte culture system and a lethal in vivo mouse infection model, integration with the in-culture and in-mouse (ICIM) pipeline enables systematic validation of pharmacodynamics, pharmacokinetics, resistance, and toxicity. This unified DFS20-based system establishes a scalable and reproducible protocol for harmonized drug efficacy evaluation across intraerythrocytic parasites and provides a foundation for the development and prioritization of pan-antiparasitic therapies.
Key features
• Common culture medium (CCM): DFS20 supports consistent Babesia and Plasmodium growth, enabling standardized, reproducible drug screening across intraerythrocytic protozoan parasites.
• Scalable antiparasitic drug screening: SYBR Green I–based in vitro assay adaptable to low-, medium-, or high-throughput compound testing in both drug-sensitive and drug-resistant strains.
• Unified red blood cell–based platform that may accelerate efforts to culture challenging or unculturable parasites, thereby expanding opportunities for drug discovery and mechanistic studies.
Keywords: Human malaria (人类疟疾)Background
The discovery of broad-spectrum antiparasitic agents requires in vitro systems that allow direct comparison of compound efficacy across multiple species under uniform conditions. However, existing intraerythrocytic parasite culture platforms rely on species-specific media with differing nutrient compositions, serum sources, and undefined protein content. These inconsistencies affect parasite growth dynamics and drug potency measurements (e.g., IC50 values), making it difficult to determine whether observed efficacy differences are due to true biological variation or culture-dependent artifacts. As a result, cross-species prioritization of chemical scaffolds with pan-apicomplexan potential remains challenging.
The clinical and epidemiological burden of intraerythrocytic apicomplexan parasites underscores the importance of developing such comparative approaches. According to the World Malaria Report 2024, there were an estimated 263 million malaria cases and approximately 597,000 deaths in 2023, with 94% of cases and 95% of deaths occurring in the WHO African Region [1]. Children under five accounted for approximately 76% of all malaria deaths, highlighting their disproportionate vulnerability [1]. Human babesiosis, caused by tick-transmitted Babesia species such as B. microti, B. duncani, B. divergens, and B. MO1, is an emerging zoonosis with increasing incidence in North America, Europe, and Asia [2–7]. According to the Centers for Disease Control and Prevention (CDC), tick-borne apicomplexan parasitic diseases have increased globally, with over 2,000 annual cases in the United States alone [8]. Resistance to first-line therapies has been documented in both genera: in Plasmodium, through mutations in PfCRT, DHFR, DHPS, and ATP4 [9–13], and in Babesia, notably in CytB and ribosomal proteins [14–17]. These shared trends of rising incidence and resistance highlight the need for new broad-spectrum antiparasitic drugs and standardized platforms that enable cross-species drug efficacy evaluation under harmonized conditions.
Plasmodium and Babesia species share key biological and pharmacological features that make cross-species drug development viable. They are obligate intraerythrocytic parasites that invade host erythrocytes using specialized cellular machineries, undergo clonal asexual replication (schizogony in Plasmodium and binary fission in Babesia), and depend on conserved metabolic pathways localized in essential organelles such as the apicoplast and mitochondrion [18–23] (Figure 1). The single mitochondrion is indispensable in both genera and serves as a validated pan-apicomplexan drug target. Mitochondrial bc1 complex inhibitors such as atovaquone and endochin-like quinolones (ELQs) exhibit potent activity against both Plasmodium and Babesia species [14,22–27]. Resistance to these agents has been linked to conserved mutations in the cytochrome b (CytB) gene in both genera, confirming shared druggable vulnerabilities and resistance trajectories [14, 17,26,28–31]. Likewise, conserved enzymes such as DHFR-TS and core mitochondrial metabolic enzymes represent shared vulnerabilities that have been pharmacologically targeted in both parasites using antifolate and glycolytic inhibitor chemotypes [18,26,32–35]. These and additional shared, yet underexplored, vulnerabilities highlight the need for a unified in vitro system for drug screening and resistance testing (Figure 1).
Despite these shared vulnerabilities, current culture methodologies are highly fragmented. P. falciparum and B. divergens are typically cultured in RPMI-1640 supplemented with human serum or lipid-based substitutes such as Albumax [36,37]. In contrast, the first in vitro culture assays for B. duncani relied exclusively on HL-1 and Claycomb media [22,26,38], both of which are proprietary and inconsistently available. A major advance occurred in 2021, when B. duncani was shown to grow continuously in human erythrocytes using DMEM/F-12 supplemented with 20% fetal bovine serum (DFS20) [39,40], providing a defined and widely accessible alternative to these media. Although B. MO1 displays growth characteristics similar to B. divergens [34], it has not yet been incorporated into a unified culture system. Of particular importance, B. duncani remains the only Babesia species for which both continuous in vitro culture in human erythrocytes and a lethal in vivo murine infection model are available [28,39–41], making it uniquely suited for translational validation of drug candidates.
To address these limitations, we evaluated DMEM/F-12 supplemented with 20% fetal bovine serum (DFS20) as a common culture medium (CCM) capable of supporting the growth of Plasmodium and Babesia. We demonstrate that DFS20 supports robust asexual replication of P. falciparum (3D7, Dd2, HB3, V1/S), B. duncani, B. divergens (Rouen 87), and B. MO1 with growth kinetics and morphology comparable to those observed in their respective traditional media [33–35]. In addition, we emphasize that DFS20 functions as a unified and practical solution for comparative drug evaluation by eliminating media-dependent variability across species. This enables consistent readouts, improves assay reproducibility, and positions DFS20 as a scalable platform for pan-apicomplexan drug screening. This unified culture system enables standardized, cross-species drug susceptibility testing and facilitates screening of compounds targeting conserved metabolic vulnerabilities. Furthermore, when integrated with the in-culture and in-mouse (ICIM) model, currently applicable only to B. duncani, it serves as a proof-of-concept for how this standardized system could support parallel assessment of pharmacodynamics, pharmacokinetics, toxicity, and resistance emergence. By replacing fragmented media systems with a single, scalable, and reproducible platform, this protocol establishes a foundation for cross-species efficacy assessment, resistance profiling, and the identification and prioritization of next-generation pan-apicomplexan therapeutics.
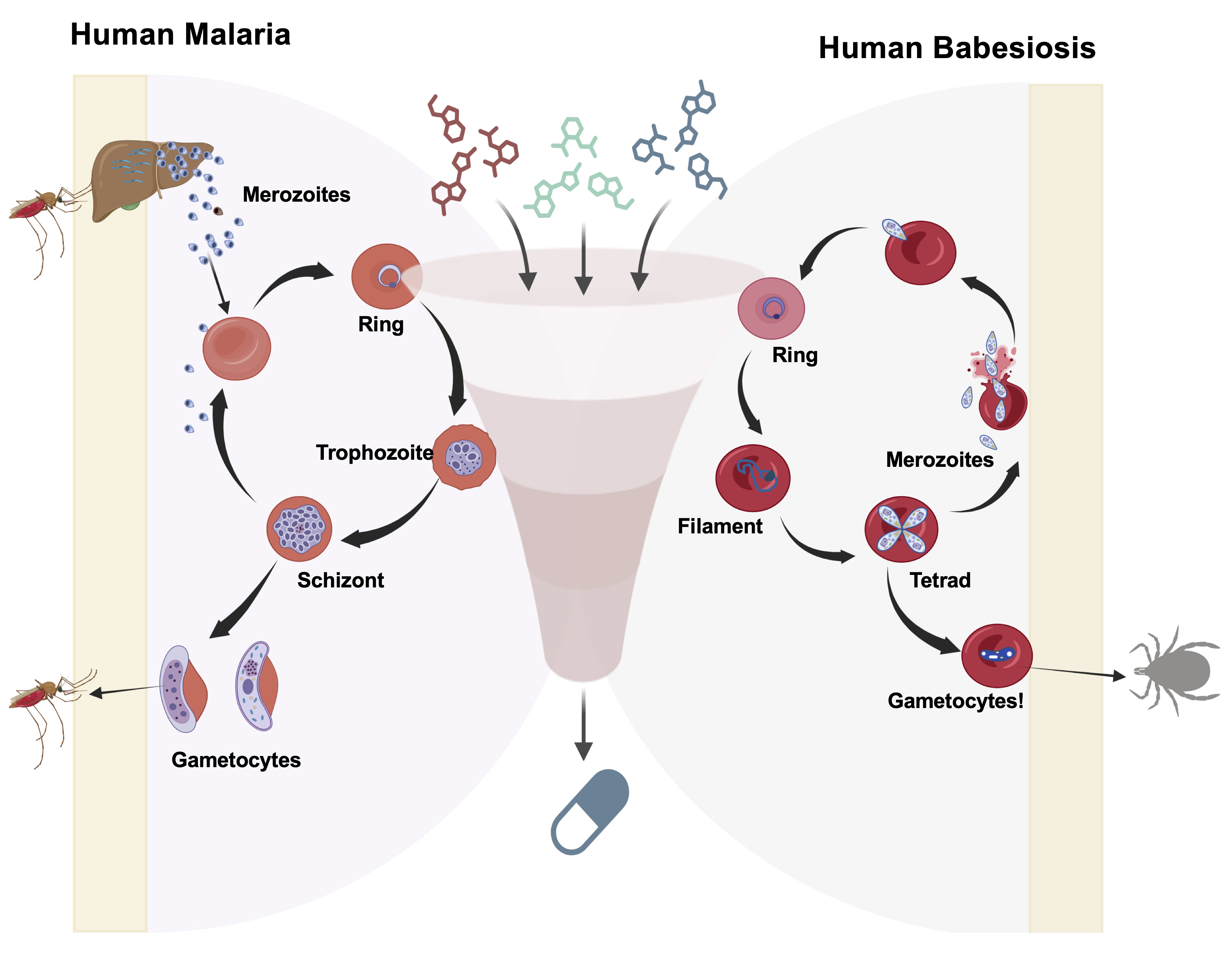
Figure 1. Schematic representation of a high-throughput screening workflow used to evaluate pan-antiparasitic drugs targeting the in vitro asexual life cycle stages of Plasmodium and Babesia parasites. The diagram illustrates the major intraerythrocytic developmental stages [ring, trophozoite, and schizont for Plasmodium falciparum (3D7, Dd2, HB1, and V1/S); ring, filament, and tetrad stages for Babesia (B. duncani, B. divergens Rouen87, and B. MO1)] and depicts merozoite invasion into fresh human red blood cells (RBCs) and continuous asexual replication. Additionally, the figure highlights the potential development of gametocytes in both parasites. A screening funnel illustrates the evaluation of diverse candidate compounds across the erythrocytic stages of malaria and babesiosis parasites.
Materials and reagents
Biological materials
1. Human packed RBCs: Human erythrocytes (Group type A+ for optimal growth) obtained from American Red Cross or Interstate Blood Bank-USA, collected in citrate phosphate dextrose (CPD), washed twice with incomplete medium, and stored at 4 °C for a maximum of 4 weeks
2. Parasite species and strains:
a. Plasmodium falciparum 3D7: (MRA-102; BEI Resources)
b. Plasmodium falciparum Dd2: (MRA-150; BEI Resources)
c. Plasmodium falciparum HB3: (MRA-1227; BEI Resources)
d. Babesia duncani WA1: (NR-12311; BEI Resources)
e. Babesia divergens Rouen87: (NR-52008; BEI Resources)
f. Babesia MO1: (NR-50441; BEI Resources)
Reagents
1. RPMI-1640 (Gibco, catalog number: 11875093); store at 4 °C
2. Albumax-II (Invitrogen, catalog number: 11021045); store at 4 °C
3. L-Glutamine (Gibco, catalog number: 25030-081); store at -20 °C
4. Hypoxanthine (Sigma, catalog number: H9636)
5. HEPES (Gibco, catalog number: 15630080); store at 4 °C
6. Sodium bicarbonate (NaHCO3) (Sigma, catalog number: S5761); store at 4 °C
7. Gentamicin (Gibco, catalog number: 15710-072)
8. DMEM-F12 (Gibco, catalog number: 11320033); store at 4 °C
9. Heat-inactivated fetal bovine serum (FBS) (Gibco, catalog number: 16140071); store at -80 °C
10. 50× HT Media Supplement Hybri-MaxTM (Sigma, catalog number: H0137); store at -20 °C
11. 100× Penicillin/Streptomycin (Gibco, catalog number: 15240-062); store at -20 °C
12. 0.008% Saponin (Sigma, catalog number: S7900)
13. 0.08% Triton X-100 (Sigma, catalog number: T8787)
14. 20 mM Tris-HCl, pH 7.5 (Sigma, catalog number: T2194)
15. 5 mM EDTA (Sigma, catalog number: E6758)
16. SYBR Green-I (Invitrogen, catalog number: S7563), diluted to 0.01% in lysis buffer; protect SYBR Green-I from light to prevent photobleaching and store at -20 °C
17. Dimethyl sulfoxide (DMSO) (Sigma, catalog number: D2650)
18. Phosphate-buffered saline (PBS) (Gibco, catalog number: 10010023)
19. Giemsa stain solution 1 (three-step fixative) (Fisher Scientific, catalog number: 22050273)
20. Giemsa stain solution 2 (three-step solution I) (Fisher Scientific, catalog number: 22050274)
21. Giemsa stain solution 3 (three-step solution II) (Fisher Scientific, catalog number: 22050275)
22. Antifolate drugs: Dihydrotriazines (51 compounds) and biguanides (28 compounds), sourced from Jacobus Pharmaceuticals, purity ≥95% (confirmed by reverse-phase HPLC)
Solutions
1. RPMI complete culture medium (see Recipes)
2. DFS20 complete culture medium (see Recipes)
3. SYBR Green-I lysis buffer (see Recipes)
Recipes
1. RPMI complete culture medium
| Reagent | Final concentration |
|---|---|
| RPMI-1640 | 99.3% |
| Albumax-II | 0.5% |
| L-Glutamine | 2 mM |
| Hypoxanthine | 50 mg/L |
| NaHCO3 | 0.225% |
| HEPES | 25 mM |
| Gentamicin | 10 mg/mL |
2. DFS20 complete culture medium
| Reagent | Final concentration |
|---|---|
| DMEM-F12 | 77% |
| Heat-inactivated FBS | 20% |
| 50× HT Media Supplement Hybri-MaxTM | 2% |
| L-Glutamine | 1% |
| Penicillin/Streptomycin | 1% |
| Gentamicin | 1% |
3. SYBR Green-I lysis buffer
| Reagent | Final concentration |
|---|---|
| Saponin | 0.008% |
| Triton X-100 | 0.08% |
| Tris-HCl, pH 7.5 | 20 mM |
| EDTA | 5 mM |
| SYBR Green-I | 0.01% |
Note: Prepare the lysis buffer in distilled water and add SYBR Green-I to a concentration of 0.01%. Protect SYBR Green-I from light to prevent photobleaching.
Equipment
1. Biological safety cabinet (Sterile GARD III), Advance biological safety cabinet (The Baker Company, catalog number: SG603)
2. CO2 incubator (Thermo Scientific, model Heracell VIOS 160i), set to 37 °C, 5% CO2, 2% O2, 93% N2 for Babesia spp. and 4% O2, 5% CO2, 91% N2 for P. falciparum
3. Sorvall legend XTR centrifuge (Thermo Scientific, catalog number: 75004521) with rotor for 15 and 50 mL tubes
4. Microscope (Nikon, model: Eclipse 5Oi)
5. Water bath (Fisher brand, model: FSGPD15D) Precision GP 10, set to 37 °C
6. Ultrasonic Bath (Branson Bransonic® M Mechanical Bath, model: M2800)
7. 96-well flat clear-bottom tissue culture plates (Corning, catalog number: 3596)
8. Greiner Bio-One Cellstar 96-well black flat bottom (Thermo Scientific, catalog number: 237108)
9. Multichannel pipettor (Eppendorf, Research Plus, 10–200 μL and 5–50 μL)
10. Microplate reader: BioTek Synergy Mx, with 480 nm excitation and 540 nm emission filters (top reading mode)
11. Vortex mixer (Scientific Industries, model: Vortex-Genie 2)
12. Sterile 15 and 50 mL conical tubes (Falcon, catalog numbers: 352096 and 352070)
13. Sterile pipette tips and pipettors (10 μL, 200 μL, 1,000 μL)
14. Sterile 1.5 mL microcentrifuge tubes (Eppendorf, catalog number: 022431081)
Software and datasets
1. Microsoft Excel, for data organization and initial calculations
2. GraphPad Prism (version 9.1.2 or advanced), for data analysis, plotting the heat maps, and sigmoidal (S) inhibition curves for IC50 determination with standard deviation (SD)
Procedure
文章信息
稿件历史记录
提交日期: Oct 30, 2025
接收日期: Jan 22, 2026
在线发布日期: Feb 24, 2026
出版日期: Mar 5, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/).
如何引用
Vydyam, P. and Ben Mamoun, C. (2026). A Standardized Culture Medium for Comparative Drug Efficacy Evaluation Across Plasmodium and Babesia Species. Bio-protocol 16(5): e5625. DOI: 10.21769/BioProtoc.5625.
分类
微生物学 > 抗微生物试验 > 杀寄生虫药试验
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link