- EN - English
- CN - 中文
Obtaining Chondroprogenitors (Articular Cartilage-Derived Cells) via Explant Methodology
采用组织块培养法获取软骨祖细胞(来源于关节软骨)
(*Contributed equally to this work, §Technical contact: d.levy@hc.fm.usp.br) 发布: 2026年03月05日第16卷第5期 DOI: 10.21769/BioProtoc.5624 浏览次数: 32
评审: Anonymous reviewer(s)
Abstract
Obtaining articular cartilage-derived cells (chondroprogenitors) by explant methodology is a reliable approach for isolating migratory progenitor cells that retain strong chondrogenic potential. This method allows cells to emerge naturally from small cartilage fragments without enzymatic digestion. The procedure consists of plating cartilage explants on a plastic surface with culture medium, from which cells subsequently migrate and adhere to the substrate. Compared with enzymatic isolation, the explant approach minimizes cellular stress and better reproduces the physiological microenvironment of cartilage tissue. This protocol can be applied to both osteoarthritic and non-osteoarthritic samples, enabling comparative studies on disease-related phenotypic differences. Overall, this technique offers a reproducible, straightforward, and minimally invasive strategy for obtaining functional chondroprogenitor cells suitable for cartilage regeneration research.
Key features
• This protocol is used to generate chondroprogenitors (articular cartilage-derived cells) from normal or osteoarthritis cartilage of the knee by explant-based isolation methodology.
• This protocol enables the isolation of chondroprogenitors without enzymatic digestion, preserving native cell–matrix interactions and minimizing chondrocyte dedifferentiation while maintaining stable chondrogenic gene expression.
• The method relies on the intrinsic migratory capacity of chondroprogenitor cells, allowing the selective enrichment of a highly proliferative and chondrogenic population.
• The method is based on a technically simple, low-cost, and scalable approach suitable for both research and translational applications.
Keywords: Chondroprogenitors (软骨祖细胞)Graphical overview
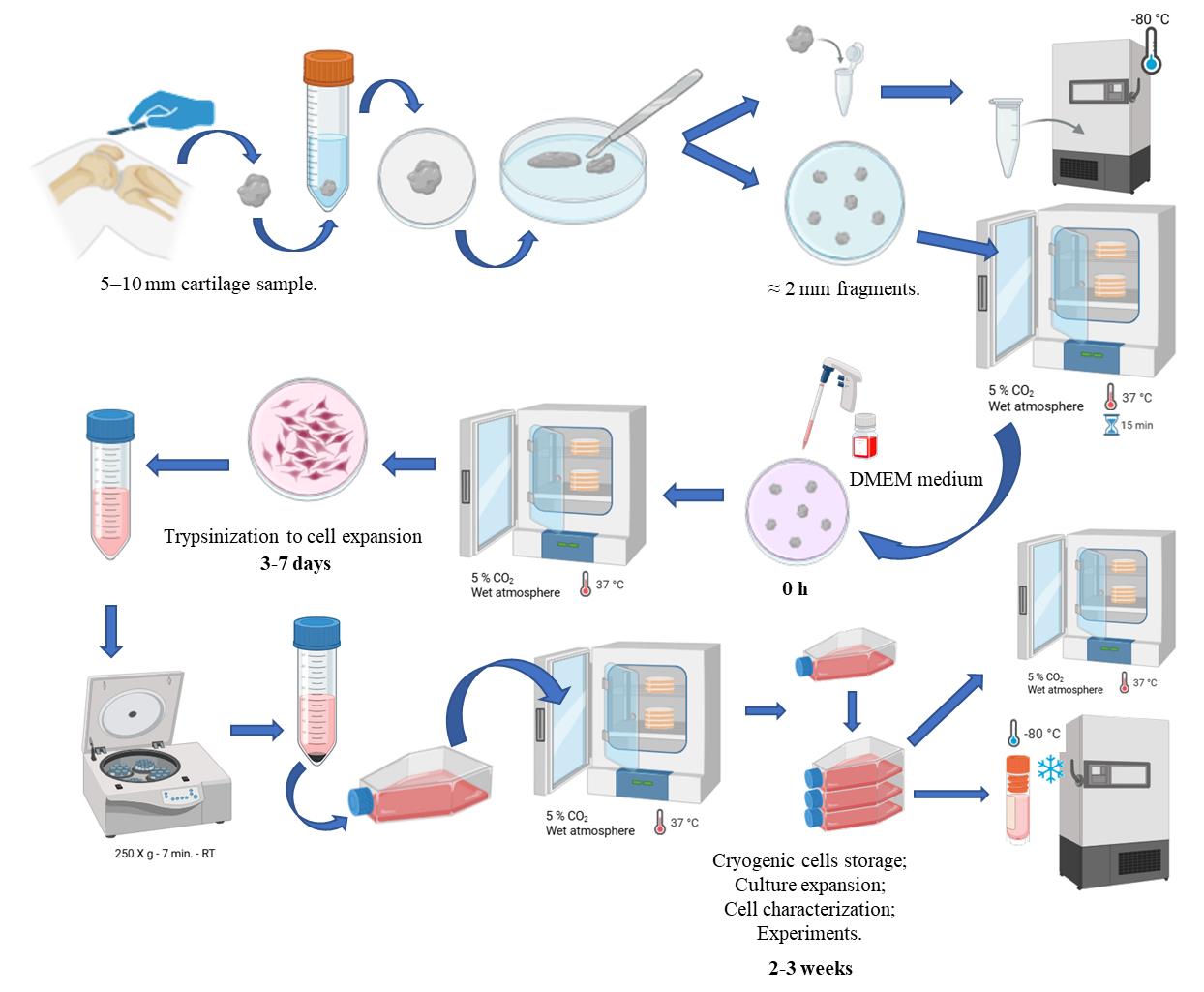
Background
Focal cartilage lesions represent a major challenge in orthopedic and knee surgery because they are highly prevalent, clinically debilitating, exhibit limited intrinsic repair capacity, and tend to progress to osteoarthritis.
The first use of cell-based therapy for cartilage repair dates to 1987 and was described in 1994 by Brittberg et al. [1], introducing autologous chondrocyte implantation (ACI). In this procedure, autologous chondrocytes harvested from the patient’s articular cartilage via arthroscopic biopsy are expanded in vitro and subsequently reimplanted into the chondral defect to promote the formation of hyaline-like repair tissue. The cultured cells are injected under a periosteal flap sutured over the defect during a second surgery. Later generations of ACI replaced the periosteum with biomaterial scaffolds, such as decellularized collagen membranes or hyaluronic acid matrices, pre-seeded with autologous cells [2]. These advances have allowed cartilage regeneration beyond the limited capacity of native tissue repair. ACI is widely recognized as an effective treatment option for focal chondral lesions, showing favorable long-term outcomes.
Progenitor cells have been identified in various adult tissues [3], including articular cartilage. Chondroprogenitors are less differentiated than mature chondrocytes and are thought to participate in cartilage regeneration, representing a promising cell source for articular cartilage repair [4]. These cells display a strong chondrogenic phenotype, low hypertrophic potential, high proliferative capacity, and maintenance of chondrogenic potential during extended in vitro expansion [5–8]. Chondroprogenitors have emerged as a promising alternative for cell-based therapy in cartilage repair. In 2004, chondroprogenitor cells were first isolated using a fibronectin adhesion assay [9]. Subsequently, alternative isolation methods were described, including migration-based selection [10] and sorting by surface markers [11–13].
The fibronectin adhesion assay selects cartilage cells that attach rapidly to fibronectin-coated substrates; it was among the first practical methods used to prospectively enrich cartilage progenitor-like cells and remains widely applied in cartilage progenitor research. This approach is relatively simple, can yield clonogenic progenitor clones, and selects based on a behaviorally relevant property (adhesion). The limitations are the heterogeneity of the cells obtained, the differing performance by cartilage source, and protocol variability [14,15].
Surface marker sorting aims to prospectively isolate chondroprogenitors by antigen expression, enabling more defined populations when robust markers are available. It presents high specificity when validated markers exist, the ability to combine markers for refined subpopulations, and high-purity populations for downstream assays [16]. As limitations, it requires cytometry infrastructure, can be time-consuming, and surface antigen expression can be contextually variable or lost during expansion [17].
The explant-based isolation of chondroprogenitor cells offers several biological and methodological advantages over enzymatic or adhesion-based protocols. In explant cultures, cells remain initially embedded within their native extracellular matrix (ECM), preserving key cell–matrix interactions that support a chondrogenic phenotype and reduce dedifferentiation typically observed during monolayer expansion [16]. Moreover, explant systems naturally select for migratory chondroprogenitors—cells capable of leaving the tissue and migrating into the surrounding environment—which may reflect an intrinsic regenerative response to cartilage injury. These cells exhibit enhanced proliferative capacity, low hypertrophic potential, and stable expression of chondrogenic genes such as SOX9, ACAN, and COL2A1 even after prolonged culture [16,18]. Furthermore, when tested in ex vivo models, chondroprogenitors isolated from explants retained high viability and regenerative capacity within platelet-rich plasma scaffolds, forming a cartilage-like matrix rich in glycosaminoglycans and type II collagen [19]. Together, these findings highlight that explant-based approaches not only preserve the native microenvironment but also enrich for a physiologically relevant progenitor population with promising potential for cartilage repair strategies. Obtaining chondroprogenitors by explant is an important way to obtain cells for both scientific research and use in ACI. Therefore, we present a protocol for obtaining chondroprogenitors by explants.
Materials and reagents
Biological materials
1. 5–10 mm full-thickness cartilage fragment from a non-weight-bearing area on the lateral edge of the trochlea or intercondylar notch
Reagents
1. DMEM low glucose (Dulbecco’s modified Eagle’s medium) (Sigma, catalog number: D5523-1L)
2. Penicillin-Streptomycin (Pen/Strep) (5,000 U/mL/5,000 μg/mL) (Thermo Fisher Scientific, Gibco, catalog number: 15070063)
3. Fetal bovine serum (FBS) (Vitrocell, catalog number: 90011)
4. 0.25% trypsin-EDTA solution (Sigma, catalog number: T4049-500mL)
5. Dimethyl sulfoxide (DMSO) (Sigma, catalog number: D-5879)
6. Sodium bicarbonate (Sigma, catalog number: S5761-1KG)
7. Trypan Blue solution (Sigma, catalog number: T8154-100ML)
8. 10× phosphate-buffered saline (PBS) pH 7.4 (Invitrogen, catalog number: AM9624)
9. Milli-Q® water (produced in-house)
10. Ethanol 70% (Vic pharma by schülke)
Solutions
1. DMEM low glucose (see Recipes)
2. DMEM complete culture medium (see Recipes)
3. PBS 1× (see Recipes)
4. Freezing medium (see Recipes)
Recipes
1. DMEM low glucose medium
| Reagent | Final concentration | Quantity |
|---|---|---|
| DMEM low glucose | 10.0 g/L | 10.0 g |
| Sodium bicarbonate | 3.7 g/L | 3.7 g |
| Milli-Q water | n/a | q.s. 1,000 mL |
Note: Adjust pH to 7.2. Filter the media in a 500 mL filter system and store at 4 °C for 6 months.
2. DMEM complete culture medium
| Reagent | Final concentration | Volume |
|---|---|---|
| DMEM low glucose medium | n/a | 445 mL |
| FBS | 10% | 50 mL |
| Pen-Strep | 50 IU/mL/50 μg/mL | 5 mL |
| Total | n/a | 500 mL |
Note: Store media at 4 °C for 3 months; before use, it must be warmed up to 37 °C in a water bath.
3. PBS 1×
| Reagent | Final concentration | Volume |
|---|---|---|
| 10× PBS | 1× | 50 mL |
| Milli-Q water | n/a | q.s. 500 mL |
Note: Adjust pH to 7.4.
4. Freezing medium
| Reagent | Final concentration | Volume |
|---|---|---|
| DMEM low glucose | n/a | 16 mL |
| FBS | 10% | 2 mL |
| DMSO | 10% | 2 mL |
| Total | n/a | 20 mL |
Note: Freezing medium should always be prepared on the day of freezing.
Laboratory supplies
1. 55 cm2 polystyrene non-treated culture dishes (Corning, catalog number: CLS430591EA)
2. Scalpel blade #11 (Descarpack, catalog number: 433-1-MKP)
3. Cell culture treated flasks, 75 cm2, vent cap (UltraCruz, Santa Cruz, catalog number: CS-200263)
4. Cell culture treated flasks, 182 cm2, vent cap (UltraCruz, Santa Cruz, catalog number: CS-200264)
5. 15 mL conical tubes (Corning, catalog number: 430791)
6. 50 mL conical tubes (Corning, catalog number: 430829)
7. Filtermax filter top fast 500 mL, pore 0.22 μm (TPP, catalog number: 9505)
8. 2 mL cryovials (Corning, catalog number: CLS431417-500EA)
9. 5 mL serological pipettes (Fisher Scientific, Fisherbrand, catalog number: 13-678-11D)
10. 10 mL serological pipettes (ExactaCruz, Santa Cruz, catalog number: SC-200281)
11. 25 mL serological pipettes (Fisher Scientific, Fisherbrand, catalog number: 13-678-11)
12. 10 μL pipette tips, filtered (Bio Point Scientific, catalog number: 321-4050)
13. 20 μL pipette tips, filtered (Bio Point Scientific, catalog number: 341-4050)
14. 200 μL pipette tips, filtered (Neptune, catalog number: BT200)
15. 1,000 μL pipette tips, filtered (Bio Point Scientific, catalog number: 361-4050)
16. Microcentrifuge tubes 0.5 mL, conical bottom (Jet Biofil, catalog number: CFT-000-005)
17. CountessTM cell counting chamber slides (Thermo Fisher Scientific, Invitrogen, catalog number: C10283)
18. BRAND® laboratory bottle, graduated with screw cap (Merck, catalog number: BR122554)
Equipment
1. Class I biological safety cabinet (VECO, catalog number: VLFS12)
2. Forma Series II Water-Jacketed CO2 Incubator (Thermo Fisher Scientific, catalog number: 3110)
3. Benchtop Centrifuge model Allegra X-12R (Beckman Coulter, catalog number: 392302)
4. Axion Vert.A1 inverted microscope (objectives 4×, 10×, 20×) (Carl Zeiss, catalog number: 431030-9010-000)
5. Micropipette Eppendorf Research plus 0.5–10 μL (Eppendorf, catalog number: 3123000020)
6. Micropipette Eppendorf Research plus 2–20 μL (Eppendorf, catalog number: 3123000039)
7. Micropipette Eppendorf Research plus 20–200 μL (Eppendorf, catalog number: 3123000055)
8. Micropipette Eppendorf Research plus 100–1,000 μL (Eppendorf, catalog number: 3123000063)
9. Scientific’s Pipet-Aid model XP (Drummond, catalog number: 4-000-101)
10. Water bath IIO2 (37 °C) (FANEM, catalog number: BM1102)
11. Countess II FL automated cell counter (Thermo Fisher Scientific, catalog number: AMQAF1000)
12. Freezer (-20 °C)
13. Ultrafreezer (Sanyo Ultra Low -80 °C, catalog number: MDF-U56VC)
14. Refrigerator (2–8 °C)
15. Lab Stirring hot plate (Thermolyne, catalog number: SPA1025B)
16. Magnetic stirring bars (Cole-Parmer Instrument Company, catalog numbers: 8551, 8550, and 8552)
17. pH/ORP/ISE Meter (Hanna Instruments, catalog number: HI3222)
18. Balance (Sartorius, catalog number: 1409)
19. Milipore Mili-Q Integral 5 Water Purification System (Milipore, catalog number: ZRXQ005BR)
20. Steam Sterilizer Nurse II (Baumer, catalog number: B0108-100)
Procedure
文章信息
稿件历史记录
提交日期: Oct 13, 2025
接收日期: Jan 26, 2026
在线发布日期: Feb 12, 2026
出版日期: Mar 5, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
如何引用
Levy, D., Giglio, P. N., De Freitas, F. A., Bydlowski, S. P. and Demange, M. K. (2026). Obtaining Chondroprogenitors (Articular Cartilage-Derived Cells) via Explant Methodology. Bio-protocol 16(5): e5624. DOI: 10.21769/BioProtoc.5624.
分类
医学
细胞生物学 > 细胞移植 > 同种异体移植
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link