- EN - English
- CN - 中文
aGPCR-HEK: A Stable High-Expression Inducible Mammalian Cell Expression System for Adhesion GPCR Structural Biology Applications
aGPCR-HEK:用于黏附型 G 蛋白偶联受体结构生物学研究的稳定高表达可诱导哺乳动物细胞表达系统
发布: 2026年03月05日第16卷第5期 DOI: 10.21769/BioProtoc.5621 浏览次数: 93
评审: David PaulAshish KabraBeatrice Li
Abstract
ADGRL4 is an adhesion G protein–coupled receptor (aGPCR) implicated in multiple tumours. In our experience, conventional insect cell-based baculovirus expression systems have not yielded sufficient correctly folded ADGRL4 protein for purification and cryo-electron microscopy (cryo-EM) analysis. Here, we describe aGPCR-HEK, a six-week protocol that establishes stable tetracycline-inducible mammalian HEK293S GnTI- TetR cell lines expressing N-terminally HA- and GFP-tagged aGPCRs. The method comprises lentiviral production in Lenti-X 293T cells, transduction of target adherent HEK293S GnTI- TetR cells, flow cytometry enrichment of uninduced GFP-positive cells displaying leaky expression, adaptation to suspension culture, and large-scale tetracycline induction and harvesting of cells for downstream purification and cryo-EM. The system yields reproducible, milligram-scale quantities of folded aGPCR suitable for structural and biochemical studies.
Key features
• Establishes a stable, tetracycline-inducible HEK293S GnTI- TetR expression system in adherent or suspension cells for high-level expression of N-terminally HA- and GFP-tagged aGPCRs.
• Uses lentiviral transduction for efficient aGPCR genomic integration and employs a tetracycline-repressor enabling high-level expression following tetracycline induction.
• Uses flow cytometry enrichment of leaky GFP-positive cells to enhance inducibility and yield.
• Provides suspension culture adaptation and large-scale tetracycline induction for structural biology-grade protein production suitable for purification and cryo-EM analysis.
Keywords: Adhesion GPCR (aGPCR) (黏附型 G 蛋白偶联受体)Graphical overview
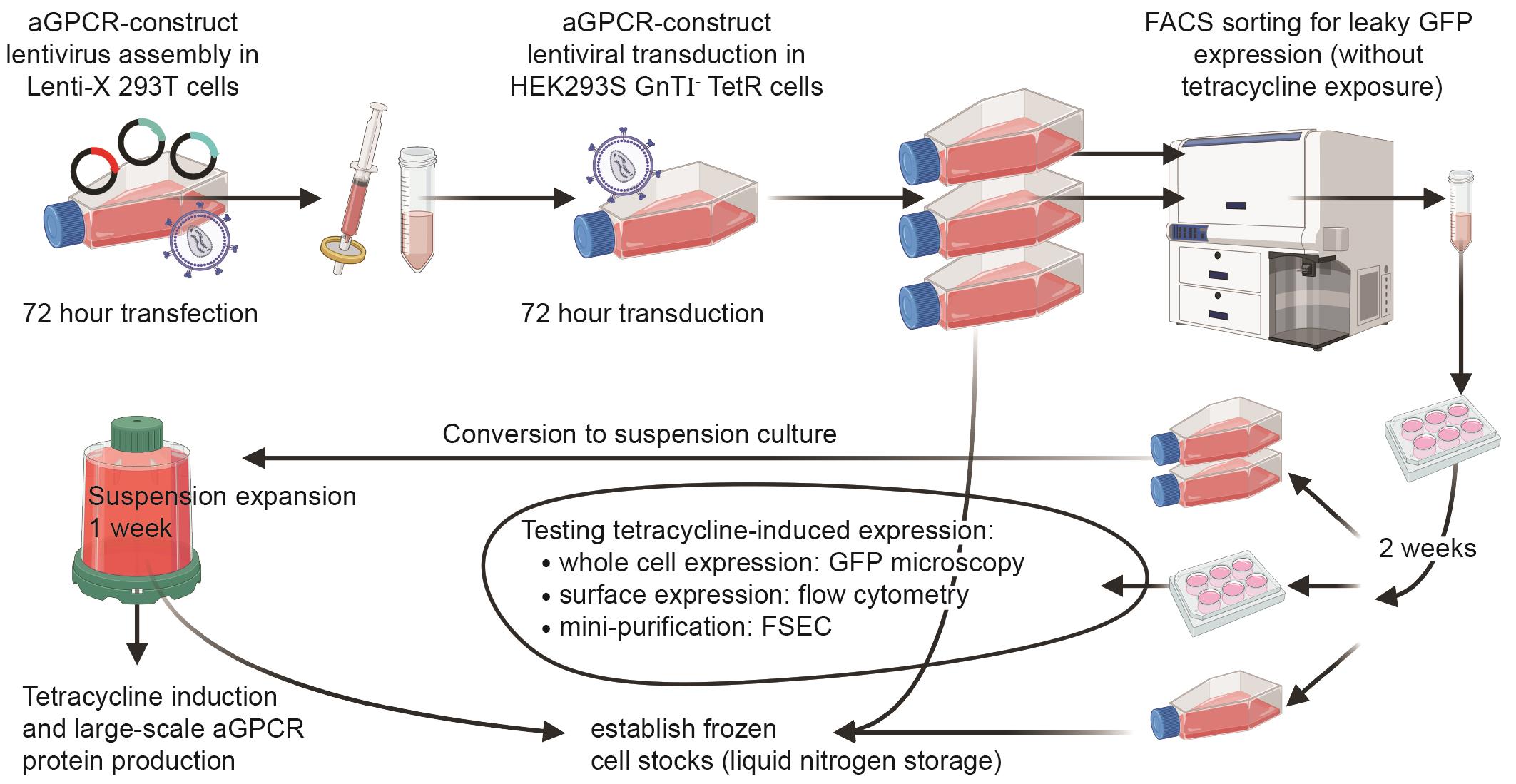
Graphical overview of the adhesion G protein–coupled receptor (aGPCR)-HEK protocol
Background
The adhesion G protein–coupled receptor (aGPCR) family comprises 32 receptors in humans that are involved in a range of important physiological and pathological processes [1]. ADGRL4 is an aGPCR that plays an important role in endothelial biology, is implicated in multiple tumour types, and is a promising therapeutic target [2–22]. Recently, we determined its high-resolution active-state structure using cryo-electron microscopy (cryo-EM) and determined that it is activated by a tethered agonist binding its orthosteric binding pocket [23]. To date, the majority of aGPCR structures determined have been expressed for large-scale protein production in insect cell-based baculovirus expression systems [24–33]. Initially, we attempted to express ADGRL4 in Sf9 (Spodoptera frugiperda) and Tni (Trichoplusia ni) insect cells using the Bac-to-Bac Baculovirus Expression System (Gibco, catalog number: 10359016). Although we were able to express ADGRL4 using this system, it did not provide sufficient high-quality folded protein for purification and cryo-EM microscopy. We thus switched to mammalian expression and developed the aGPCR-HEK protocol. This was successfully employed to express correctly folded ADGRL4 protein at a large scale for structural studies, allowing us to determine the first high-resolution structure of ADGRL4 [23].
Here, we present aGPCR-HEK, a six-week protocol that establishes a stable tetracycline-inducible mammalian HEK293S GnT1- TetR cell line expressing an N-terminally HA- and GFP-tagged aGPCR. This method consists of (i) lentiviral production in Lenti-X 293T cells, (ii) transduction of target adherent HEK293S GnT1- TetR cells, (iii) flow cytometry–positive selection of uninduced GFP-positive HEK293S GnTI- TetR cells (selecting for cells exhibiting leaky low expression of the insert in the absence of tetracycline), (iv) adaptation from adherent to suspension cell culture, and (v) large-scale tetracycline or doxycycline induction for downstream protein purification and cryo-EM. A stable inducible cell line offers superior expression and scalability over transient transfection and minimises the formation of intracellular, misfolded, inactive receptor [34]. Using the aGPCR-HEK protocol, we purified approximately 250 μg of high-quality CTF2B (the unstable active-state version of ADGRL4) from 2 L of HEK293S GnTI- TetR cells [23]. CTF2B denotes the constitutively active C-terminal fragment version of ADGRL4 (residues T407-R690) fused to a C-terminally tethered mini-Gq (βγ-binding “version B”), which stabilises the active-state receptor–G protein complex for downstream purification and cryo-EM. This yield was sufficient for determining the active-state high-resolution structure. The yield is significantly higher when expressing full-length inactive aGPCR receptors, as they are more stable. The ADGRL4 purification protocol for cryo-EM is described in a companion Bio-protocol manuscript [35].
The second-generation lentiviral system used in this method is derived from HIV-1 and has been engineered to be replication-incompetent [36,37]. It comprises three plasmids: (i) a packaging plasmid (psPAX2), (ii) an envelope plasmid (pMD2.G), and (iii) a transfer plasmid. For the transfer plasmid, we modified the pHR-CMV-TetO2_3C-Twin-Strep transfer plasmid [38] by deleting its 3C and Twin Strep sequences.
Lenti-X 293T cells were chosen for lentivirus production due to their high-level expression of lentivirus. Viral transduction was then performed on HEK293S GnTI- TetR cells. HEK293S GnTI- TetR cells are HEK293S cells lacking the N-acetylglucosaminyltransferase I (GnTI) enzyme encoded by MGAT1 [39,40]. GnTI performs the first step in complex N-glycan processing within the Golgi and, without its glycans, cannot be converted into complex, heterogeneous sugar chains. This ensures that GnTI- cells produce glycoproteins with minimal glycan heterogeneity. The HEK293S GnTI- cell line and its later TetR inducible derivative cell line were originally developed for X-ray crystallography because the presence of complex heterogeneous N-glycans hinders crystal packing and X-ray diffraction [39,40]. Improved homogeneity of N-glycosylated proteins expressed in GnTI- cells makes interpretation of western blots easier because discrete bands are observed rather than a smear produced in wild-type HEK293S cells.
During its original development, the HEK293S GnTI- TetR cell line was stably transfected under blasticidin selection with a pcDNA6/TR plasmid [39,40], integrating a tetracycline repressor (TetR) [41] under constitutive CMV promoter expression as well as a blasticidin resistance gene. HEK293S GnTI - TetR cells thus require culture in medium supplemented with 5 μg/mL blasticidin. This protocol is also applicable to using the T-REx system (Invitrogen, catalog number: K102001) with full-length N-glycans.
In the absence of tetracycline or doxycycline, TetR binds to tetracycline operator (TetO) sequences present within the tetracycline response element (TRE) downstream of the transgene’s CMV promoter. This represses transcription of the aGPCR transgene. When tetracycline or doxycycline is present, they bind to TetR, causing a conformational change that prevents TetR from binding TetO sites. This releases the promoter from repression and permits robust transgene expression. A small subset of transduced HEK293S GnTI- TetR cells displays low-level leaky transgene expression in the absence of tetracycline or doxycycline [42]. Because these cells typically produce higher transgene levels upon induction than non-leaky cells, they are preferentially selected in the aGPCR-HEK protocol by GFP-based fluorescence-activated cell sorting (FACS) [43,44].
Finally, all procedures involving lentivirus must be conducted within a Class II microbiological safety cabinet according to institutional and national biosafety guidelines. Personnel must wear appropriate protective equipment (laboratory coat, gloves, eye protection), and work surfaces and equipment must be decontaminated with a virucidal disinfectant (e.g., Virkon) after handling lentiviral material. All liquids and materials potentially contaminated with lentivirus should be inactivated and disposed of according to institutional and national biosafety rules.
Materials and reagents
Biological materials
1. Lenti-X 293T cell line (Takara, catalog number: 632180); growth medium: DMEM/F-12 with 10% tetracycline-free FBS
2. HEK293S GnTI- TetR cell line (Dr Philip J Reeves [39,40], University of Essex, UK); growth medium: DMEM with 10% tetracycline-free FBS and 5 μg/mL blasticidin.
Note: The aGPCR-HEK protocol is also applicable when using the T-REx expression system with full-length N-glycans (Invitrogen, catalog number: K102001); growth medium: DMEM with 10% tetracycline-free FBS and 5 μg/mL blasticidin.
3. Lentivirus packaging plasmids:
a. psPAX2 packaging plasmid (Addgene, catalog number: 12260)
b. pMD2.G envelope plasmid (Addgene, catalog number: 12259)
c. pHR-CMV-TetO2 transfer plasmid; generated by deleting the 3C-Twin-Strep sequences (bases 8294-8419) from the pHR-CMV-TetO2_3C-Twin-Strep plasmid [38] (Addgene, catalog number: 113883)
4. Adhesion GPCR construct:
a. ADGRL4 CTF2B (C-terminal fragment version 2B [23]: ADGRL4 with N-terminal HA-, GFP-, and purification tags) in pcDNA3.1 plasmid; the CTF2B ADGRL4 sequence was derived from the ADGRL4 Uniprot reference sequence (Q9HBW9) and comprises amino acids T407-R690
Reagents
1. Dulbecco's modified Eagle medium (DMEM) with GlutaMAX supplement, 500 mL (Gibco, catalog number: 31966-021); store at 4 °C
2. DMEM/F-12 medium, 500 mL (Gibco, catalog number: 11330-032); store at 4 °C
3. FreeStyle 293 expression medium, 1,000 mL (Gibco, catalog number: 12338-018); store at 4 °C
4. Fetal bovine serum (FBS) tetracycline-free, 500 mL (Biosera, catalog number: FB-1001T/500); store at -20 °C
5. Phosphate-buffered saline (PBS), pH 7.4, 500 mL (Gibco, catalog number: 10010015)
6. TrypLE Express enzyme, 100 mL (Gibco, catalog number: 12604013); store at 4 °C
7. Lipofectamine LTX reagent with Plus reagent, 0.3 mL (Invitrogen, catalog number: 15338030); store at 4 °C
8. Opti-MEM reduced-serum medium, 500 mL (Gibco, catalog number: 11058021); store at 4 °C
9. Blasticidin S HCl (10 mg/mL stock), 10 mL (Thermo Scientific, catalog number: A1113903); store at -20 °C
10. Polybrene (10 mg/mL stock), 1 mL (Sigma-Aldrich, catalog number: TR-1003-G); store at -20 °C
11. Lenti-X GoStix Plus, 20 tests (Takara, catalog number: 631280)
12. Dimethyl sulfoxide (DMSO), 100 mL (Sigma-Aldrich, catalog number: 34869)
13. Virkon disinfectant virucidal tablets (Day Impex Ltd, catalog number: 330013)
14. Tetracycline 98%–102% (HPLC) 25 g (Sigma-Aldrich, catalog number: T3258-25G); store at -20 °C
15. Absolute ethanol, 2.5 L (Sigma-Aldrich, catalog number: 32221-2.5L-M)
16. Trypan blue solution 0.4%, 100 mL, 2.5 L (Gibo, catalog number: 15250061)
17. Anti-HA monoclonal antibody conjugated to allophycocyanin (APC), 200 μL (Miltenyi Biotec, catalog number: 130-123-553); store at 4 °C
Solutions
1. DMEM supplemented with 10% tetracycline-free FBS (see Recipes)
2. DMEM supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin (see Recipes)
3. DMEM/F-12 supplemented with 10% tetracycline-free FBS (see Recipes)
4. FreeStyle 293 supplemented with 1% tetracycline-free FBS and 5 μg/mL blasticidin (see Recipes)
5. Flow cytometry buffer (see Recipes)
6. FACS catch buffer (see Recipes)
7. Cryopreservation medium (see Recipes)
8. 1% Virkon disinfectant solution (see Recipes)
9. Tetracycline solution (see Recipes)
Recipes
Note: All FBS used should be tetracycline-free. All recipes should be prepared under sterile conditions except for Virkon disinfectant solution.
1. DMEM supplemented with 10% tetracycline-free FBS
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM medium | 90% | 450 mL |
| Tetracycline-free FBS | 10% | 50 mL |
| Total | n/a | 500 mL |
Store at 4 °C.
2. DMEM supplemented with 10% tetracycline-free FBS and blasticidin
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM medium | 90% | 449.75 mL |
| Tetracycline-free FBS | 10% | 50 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 250 μL |
| Total | n/a | 500 mL |
Store at 4 °C.
3. DMEM/F-12 supplemented with 10% tetracycline-free FBS
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM/F-12 medium | 90% | 450 mL |
| Tetracycline-free FBS | 10% | 50 mL |
| Total | n/a | 500 mL |
Store at 4 °C.
4. FreeStyle 293 supplemented with 1% tetracycline-free FBS and blasticidin
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| FreeStyle 293 medium | 99% | 989.5 mL |
| Tetracycline-free FBS | 1% | 10 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 500 μL |
| Total | n/a | 1,000 mL |
Store at 4 °C.
5. Flow cytometry buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| PBS | 98% | 19.6 mL |
| Tetracycline-free FBS | 2% | 0.4 mL |
| Total | n/a | 20 mL |
Store at 4 °C. Keep on ice when in use.
6. FACS catch buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM | 90% | 17.99 mL |
| Tetracycline-free FBS | 10% | 2 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 10 μL |
| Total | n/a | 20 mL |
Store at 4 °C. Keep on ice when in use.
7. Cryopreservation buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM | 70% | 6.995 mL |
| Tetracycline-free FBS | 20% | 2 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 5 μL |
| DMSO | 10% | 1 mL |
| Total | n/a | 10 mL |
Make up fresh before use.
8. 1% Virkon disinfectant solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Tap water | 99% | to a total volume of 500 mL |
| Volume of lentivirus-rich discarded cultured medium | × mL | |
| Virkon disinfectant virucidal tablet | 1% | Single 5 g tablet |
| Total | n/a | 500 mL |
9. Tetracycline stock solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Tetracycline powder | 1 mg/mL | 20 mg |
| Absolute ethanol | ~99.9% | 20 mL |
| Total | n/a | 20 mL |
a. Weigh 20 mg of tetracycline powder (~100% HPLC purity) on an analytical balance using a weighing boat or paper.
b. Add the measured tetracycline to ~15 mL of absolute ethanol (≥95%) in a foil-wrapped 50 mL centrifuge tube. Gently mix until dissolved and then bring to 20 mL with further absolute ethanol.
c. To ensure sterility, filter through a 0.22 μm PES filter and decant into a new foil-wrapped sterile 50 mL centrifuge tube.
d. Under sterile conditions, aliquot as appropriate into foil-covered tubes and protect from light (tetracycline is light sensitive). Store at -20 °C for up to a year.
e. When inducing HEK293S GnTI- TetR cells, the final concentration of tetracycline should be 1 μg/mL.
Laboratory supplies
1. Fisherbrand sterile PES syringe filters, 0.45 μm pore size (Fisher Scientific, catalog number: 15216869)
2. Fisherbrand sterile PES syringe filters, 0.2 μm pore size (Fisher Scientific, catalog number: 15206869)
3. CellTrics 50 μm filters (CellTrics, catalog number: 04-004-2327)
4. Sarstedt sterile 5 mL polypropylene tube (L × Ø): 75 × 12 mm (Sarstedt, catalog number: 55.526.006)
5. Falcon 5 mL sterile round-bottom polypropylene test tube with snap cap (Corning, catalog number: 352063)
6. 50 mL sterile Luer Lock syringes, pack of 25 (Terumo, catalog number: 8SS50L1)
7. 96-well round (U) bottom plates (Thermo Scientific, catalog number: 163320)
8. 50 mL centrifuge tubes (Corning, catalog number: CLS430828-100EA)
9. 6-well CytoOne plates, TC-treated (Starlab, catalog number: CC7682-7506)
10. 24-well CytoOne plates, TC-treated (Starlab, catalog number: CC7682-7524)
11. Falcon T75 cm2 rectangular canted neck cell culture flasks with vented caps (Corning, catalog number: 353136)
12. Falcon T175 cm2 rectangular straight neck cell culture flasks with vented caps (Corning, catalog number: 353112)
13. Corning 500 mL polycarbonate Erlenmeyer flasks with flat cap (Corning, catalog number: 430422)
14. Corning 125 mL polycarbonate Erlenmeyer flask with flat cap (Corning, catalog number: 430421)
15. Corning 850 cm2 polystyrene roller bottle with easy grip cap (Corning, catalog number: 430849)
16. Corning 1.2 mL cryogenic vial (Corning, catalog number: 430487)
17. Mr. Frosty freezing container (Thermo Scientific, catalog number: 51000001)
Equipment
1. Class II biological safety cabinet: LaboGene Mars 1200 Class II Biological Safety Cabinet (LaboGene, catalog number: Mars 1200)
2. Stationary incubator: Sanyo MCO-18AIC CO2 incubator (Sanyo, catalog number: MCO-18AIC)
3. Shaking incubator: Infors HT Multitron Pro Triple Stacked Incubator Shaker (Infors, catalog number: Multitron Pro); shaking incubator (37 °C; 8% CO2; >95% humidity, shaking speed of 125 rpm)
4. Temperature-controlled 1.5 mL tube centrifuge: Eppendorf 5418 R Centrifuge (Eppendorf, catalog number: 5418 R)
5. Temperature-controlled 15–50 mL tube swinging-bucket centrifuge: Beckman Coulter Allegra X-12R Refrigerated Centrifuge (Beckman Coulter, catalog number: X-12R)
6. Temperature-controlled floor-standing centrifuge for 1 L bottles: Beckman Coulter Avanti JXN-26 Refrigerated Centrifuge (Beckman Coulter, catalog number: JXN-26)
7. Microscope (brightfield and fluorescence): Life Technologies EVOS FL microscope (Life Technologies, catalog number: AMF4300)
8. FACs cell sorter: Invitrogen Bigfoot Cell Sorter (Invitrogen, catalog number: PL00304)
9. Flow cytometer (Sony, model: ID7000 Spectral Cell Analyzer)
10. FSEC system: Shimadzu HPLC system comprising the following components: Shimadzu Degasser Module (DGU-20A), Shimadzu Solvent Delivery Module (LC-20AD), Shimadzu Autosampler Module (SIL-20AC), Shimadzu Column Oven Module (CTO-20AC), Shimadzu UV-detector Module (SPD-20A), Shimadzu Fluorescence Detector Module (RF-20A), Shimadzu Fraction Collector Module (FRC-40); HPLC guard column: Agilent Bio SEC-5 guard column (300 Å, 7.8 × 50 mm, 5 μm) (Agilent, catalog number: 5190-2530), HPLC column: Agilent Bio SEC-5 column (300 Å 4.6 × 300 mm 5 μm) (Agilent, catalog number: 5190-2528)
Software and datasets
1. Microsoft Excel (Microsoft, version 16.101.3 (25100321)) for calculations
2. FlowJo (Becton Dickinson & Company, version 10.10.0) for flow cytometry data analysis
Procedure
文章信息
稿件历史记录
提交日期: Dec 9, 2025
接收日期: Jan 28, 2026
在线发布日期: Feb 10, 2026
出版日期: Mar 5, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
如何引用
Favara, D. M. and Tate, C. G. (2026). aGPCR-HEK: A Stable High-Expression Inducible Mammalian Cell Expression System for Adhesion GPCR Structural Biology Applications. Bio-protocol 16(5): e5621. DOI: 10.21769/BioProtoc.5621.
分类
生物工程
分子生物学 > 蛋白质 > 表达
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link